How much pO 2 would it take in an extremely CO 2 -rich environment to saturate hemoglobin
Question:
How much pO2 would it take in an extremely CO2-rich environment to saturate hemoglobin 90 percent?
(A) 15
(B) 30
(C) 45
(D) 60
The affinity of hemoglobin for oxygen is reduced by many factors, including low pH and high CO2. The graph below shows the different dissociation curves that maternal (normal) hemoglobin and fetal hemoglobin have.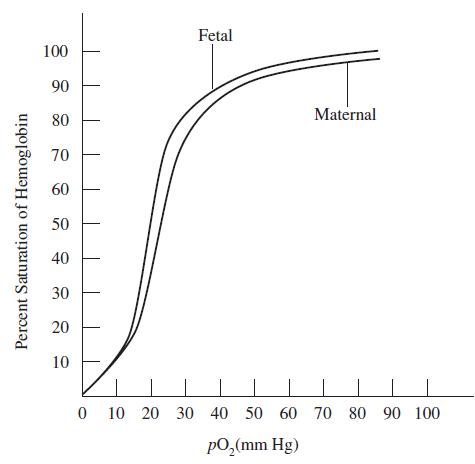
Transcribed Image Text:
Percent Saturation of Hemoglobin 100 90 80 70 60 50 40 30 20 10 Fetal Maternal 0 10 20 30 40 50 60 70 80 90 100 pO₂(mm Hg)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
D The CO 2 rich environment would decrease the af...View the full answer

Answered By

Diane Joyce Pastorin
Please accept my enthusiastic application to solutioninn. I would love the opportunity to be a hardworking, passionate member of your tutoring program. As soon as I read the description of the program, I knew I was a well-qualified candidate for the position.
I have extensive tutoring experience in a variety of fields. I have tutored in English as well as Calculus. I have helped students learn to analyze literature, write essays, understand historical events, and graph parabolas. Your program requires that tutors be able to assist students in multiple subjects, and my experience would allow me to do just that.
You also state in your job posting that you require tutors that can work with students of all ages. As a summer camp counselor, I have experience working with preschool and kindergarten-age students. I have also tutored middle school students in reading, as well as college and high school students. Through these tutoring and counseling positions, I have learned how to best teach each age group.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

The Princeton Review AP Biology Premium Prep 2023
ISBN: 9780593450659
2023 Edition
Authors: The Princeton Review
Question Posted:
Students also viewed these Sciences questions
-
Based on the graph, it can be concluded that (A) fetal hemoglobin surrenders O 2 more readily than maternal hemoglobin (B) the dissociation curve of fetal hemoglobin is to the right of maternal...
-
Which of the following processes would likely shift the normal dissociation curve to the right? (A) Photosynthesis (B) Respiration (C) Fermentation (D) Mitosis The affinity of hemoglobin for oxygen...
-
Hemoglobins affinity for O 2 (A) Decreases as blood pH decreases (B) Increases as H + concentration increases (C) Increases as blood pH decreases (D) Decreases as OH concentration increases The...
-
Problem A (20 points): Loco Farms Company sold 36,000 units of its only product and incurred a $18,672 loss (ignoring taxes) for the current year as shown here. During a planning session for year...
-
Texas International Speedway, Inc. (TIS) filed a registration statement for $4,398,900 in securities with the proceeds to be used to construct an automobile racetrack called the Texas International...
-
How do you estimate the potential margin and capital turnover for a young, high-growth company? Are the companys current margin and capital turnover relevant?
-
The Arcadia Company was founded by Thomas Smith and began operations in July 2015 as a neighborhood hardware store, selling branded lawn and garden supplies, household products, and small tools. The...
-
In October 2010, Tibbetts Company had the following production and cost data: Beginning inventory units (80% complete as to DM; 45% complete as to DL; 30% complete as to OH) ....... 42,600 October...
-
Use the transforms in the table below to find the inverse Laplace transform of the following function. F(s)= 9 Click the icon to view the table of Laplace transforms. The inverse Laplace transform of...
-
How many unique, non-homologous chromosomes are present in this species? (A) 4 (B) 8 (C) 16 (D) 32 The somatic cells in a newly identified sexually reproducing species are found to be octoploidy...
-
Which of the following best describes the fish population in the year 1958? (A) All medium-orange in color (B) Mostly medium-orange fish with some nearly white and some nearly brown (C) Mostly white...
-
Define a line integral of a function along a smooth curve C in the plane and in space. How do you evaluate the line integral as a definite integral?
-
What is a verified pleading?
-
What three elements must be included in the caption of a complaint?
-
How is the California court system structured?
-
What is civil litigation and how does it differ from other types of litigation?
-
What ethical standards paralegals must follow?
-
The table below appeared in Exercise 6. It shows the type of business and the gender of the CEO for your firms five major clients: You plan to randomly choose one company from the group. Find the...
-
Phosgene, COCl2, is a toxic gas used in the manufacture of urethane plastics. The gas dissociates at high temperature. At 400oC, the equilibrium constant Kc is 8.05 104. Find the percentage of...
-
Quantity A Quantity B r p + q 1 Quantity A is greater. Quantity B is greater. The two quantities are equal. The relationship cannot be determined...
-
Quantity A Quantity B x 70 Quantity A is greater. Quantity B is greater. The two quantities are equal. The relationship cannot be determined from the...
-
In the figure above, if AB is parallel to CD, then ABD = 11 22 33 46 134 A 44 E 2x' 3x B
-
Question 1 1. Calculate the effective interest rate for monthly compounding when the annual interest rate is 10%. 2. Define 'LCOE (levelized cost of electricity)'. 3. Determine the LCOE for the plant...
-
Maga Rehab Hospital, a not-for-profit corporation, is estimating its corporate cost of capital. Its tax-exempt debt currently requires an interest rate of 9.5 percent, and its target capital...
-
(chi-square) - We will be using the chapek9 data set on jamovi. Test the research hypothesis that, robots prefer data and humans prefer puppies (**note** start with adding the flowing filter: fx =...

Study smarter with the SolutionInn App


