Tran?s alkenes are converted into their cis isomers and vice versa on epoxidation followed by treatment of
Question:
Tran?s alkenes are converted into their cis isomers and vice versa on epoxidation followed by treatment of the epoxide with triphenylphosphine. Propose a mechanism for the epoxide ? alkene reaction.
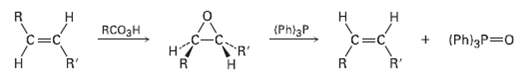
Transcribed Image Text:
н RCO3H (Phl3P R' (Ph)3P 0 на R' R'
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 41% (12 reviews)
Ph3P H Ph3P H rotate R 180 R H Ph3P rotatio...View the full answer

Answered By

Wahome Michael
I am a CPA finalist and a graduate in Bachelor of commerce. I am a full time writer with 4 years experience in academic writing (essays, Thesis, dissertation and research). I am also a full time writer which assures you of my quality, deep knowledge of your task requirement and timeliness. Assign me your task and you shall have the best.
Thanks in advance
4.90+
63+ Reviews
132+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the following reaction (remember to use curved arrows when showing a mechanism): CH3CHCH-CH-OH CH,C-CH, CH,CHCH-CH-OCCH, + CH CH
-
Propose a mechanism for the following reaction that explains why the configuration of the asymmetric carbon in the reactant is retained in the product: COO NaNO2 HCl NH2 COO
-
Propose a mechanism for reaction of the first three propylene units in the polymerization of propylene in the presence of a peroxide. ROOR- H2C CHCH high pressure C-C propylene I7 polypropylene
-
Cubitene is a diterpene present in the defense secretion of a species of African termite. What unusual feature characterizes the joining of isoprene units in cubitene?
-
A procedural leader sets an agenda, makes sure that everyone knows what's due for the next meeting, and checks to be sure that tasks are carried out. Does Malcom fulfill the role of an effective...
-
Perry Co. predicts it will use 25,000 units of material during the year. The expected daily usage is 200 units, and there is an expected lead time of five days and a desired safety stock of 500...
-
6. YOU BE THE JUDGE WRITING PROBLEM Johnny Carson was for many years the star of a well-known television program, The Tonight Show. For about 20 years, he was introduced nightly on the show with the...
-
(Conversion of Bonds) On January 1, 2010, when its $30 par value common stock was selling for $80 per share, Bartz Corp. issued $10,000,000 of 8% convertible debentures due in 20 years. The...
-
Avitech has a$10.0million debt issue outstanding, with a6.0%coupon rate. The debt has semi-annual coupons, the next coupon isdue in six months, and the debt matures in five years. It iscurrently 2...
-
A key aspect of H&M's strategy is to provide value to customers by maximizing perceived product benefits, minimizing prices, or both. Given this, how can H&M further increase the value of its...
-
In light of ?our answer to Problem 19.56, propose a mechanism for the formation of 3, 5 -dimethylisoxazole from hydroxylamine and 2, 4-pentanedione. CH 3,5-Dimethylisoxazole
-
Treatment of an ?, ? unsaturated ketone with basic aqueous hydrogen peroxide yields an epoxy ketone. The reaction is specific to unsaturated ketones; isolated alkene double bonds do not react....
-
Homestead Telephone was formed in the 1940s to bring telephone services to remote areas of the U.S. Midwest. The early equipment was quite primitive by today's standards. All calls were handled...
-
You are analyzing General Electric's common stock as a potential investment. You estimate that GE's dividends will grow at an average rate of 5.5% per year over the long term. GE's last dividend, (we...
-
If Erika's marginal tax rate in 2023 is 32%, and her expected marginal tax rate at retirement in 25 years is 10%, should she choose to contribute to her employer's traditional 401(k) or Roth 401(k)?
-
Will the following programs terminate? (a) int balance while (true) { 10; if (balance < 9) break; balance balance 9; } System.out.println("Balance is " (b) + balance); int balance = 10; while (true)...
-
Martha's Vineyard Wine Shop has a debt cost rate of 6 percent and an equity cost of 15 percent. The firm is trying to decide how to calculate its weighted average cost of capital in order to decide...
-
Problem 2: Income statement Adjusted trial balances of accounts Research and development Unrealized gain on available for sale securities Restructuring costs Cost of goods sold 700,000 Interest...
-
The General Social Survey asked 46,349 women how many children they had. The results were as follows. a. What is the probability that a sampled woman has two children? b. What is the probability that...
-
What is a manufacturing system?
-
One of the chemical reactions that occurs in the formation of photochemical smog is O 3 + NO NO 2 + O 2 . Estimate r H for this reaction by using appropriate Lewis structures and data from Table...
-
Give common or systematic names for each of the following compounds: (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) CH3N+H3 CH3CO2 - (l) (m) (n) NH2 HIN NH2 CH3 NH2 OCH NIH N NH2 SO NH2 NH2 CH3
-
Which is the most basic nitrogen in each compound. Explain your choices. (a) (b) (c) NH2 HN
-
Show how you might prepare benzylamine from each of the following compounds: (a) (b) (c) Benzyl bromide (two ways) (d) Benzyl tosylate (e) Benzaldehyde (f) Phenylnitromethane (g) NH2 Benzylamine CN...
-
If a price floor is above equilibrium, we may see: Question 8 options: a ) a low quality of the good being offered b ) a shortage of the good or service in question c ) no impact on the market d )...
-
5. Explain 4 indications for thyroidectomy. (4 Marks) 6. State three advantages and three disadvantages of ambulatory surgery. (6 marks)
-
1. Describe at least FOUR post-operative complications. 2. State six elements of perioperative surgical asepsis.

Study smarter with the SolutionInn App


