Treatment of an ?, ? unsaturated ketone with basic aqueous hydrogen peroxide yields an epoxy ketone. The
Question:
Treatment of an ?, ? unsaturated ketone with basic aqueous hydrogen peroxide yields an epoxy ketone. The reaction is specific to unsaturated ketones; isolated alkene double bonds do not react. Propose a mechanism.
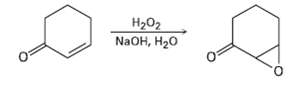
На02 NaOH, H20 O.
Step by Step Answer:
HOOH OH Hydrogen peroxide and hydroxide react to form water and ...View the full answer



Related Video
Hydrogen peroxide can be used as a mild antiseptic to curb superficial skin infections such as athlete’s foot, but only in diluted quantities. To combat stinky feet, try soaking your feet in a solution of 1 part 3% hydrogen peroxide and 3 parts warm water for 15-20 minutes, then drying them thoroughly. This will kill odor-causing bacteria and soften your feet. To treat athlete\'s foot, you can use a similar solution, but only in diluted quantities, and soak your feet for 30 minutes. Hydrogen peroxide can also be used to keep vegetables fresh by adding 1/4 cup to a bowl of cold water, soaking the vegetables for 20-30 minutes, then draining, drying, and refrigerating them. Alternatively, you can spray vegetables with a solution of 3% hydrogen peroxide and let them stand for a few minutes before rinsing and drying. To keep leftover salad fresh, spray it with a solution of 1/2 cup water and 1 Tbsp. 3% hydrogen peroxide, drain, cover, and refrigerate.
Students also viewed these Organic Chemistry questions
-
Hydrogen peroxide in aqueous solution decomposes by a first-order reaction to water and oxygen. The rate constant for this decomposition is 7.40 104/s. What quantity of heat energy is initially...
-
Hydrogen peroxide and ferrous sulfate react to produce hydroxyl radical (HO), as reported in 1894 by English chemist H. J. H. Fenton. When tert-butyl alcohol is treated with HO generated this way, it...
-
Propose a mechanism for the reaction of cyclohexyl methyl ketone with excess bromine in the presence of sodium hydroxide.
-
Write a detailed executive summary about the appropriation and advancement of Information and Communication Technology (ICT)
-
Groupthink is the tendency for teams to put such a high premium on agreement that directly (or indirectly) punish dissent. Was the Schumacker's team guilty of groupthink? Why or why not?
-
Lopez Chemical Inc. requires 20,000 gallons of material annually; the cost of placing an order is $20; the annual carrying cost per gallon is $5. Required: Determine the EOQ from potential order...
-
5. Caudle worked at Betts Lincoln-Mercury dealer. During an office party, many of the employees, including President Betts, were playing with an electric auto condenser, which gave a slight electric...
-
The following events took place for Chaterjee Manufacturing Company during January 2014, the first month of its operations as a producer of digital thermometers: a. Purchased $ 95,200 of materials....
-
Suppose you invest equal amounts in a portfolio with an expectedreturn of 20% and a standard deviation of returns of 16% and arisk-free asset with an interest rate of 4%; calculate the standarddevi 2...
-
Mohamad, owner of an residential furnished apartment's in Dubai, performed an economic analysis 4 years ago when he decided to place an new eefficient central AC unit for each apartments instead of...
-
Tran?s alkenes are converted into their cis isomers and vice versa on epoxidation followed by treatment of the epoxide with triphenylphosphine. Propose a mechanism for the epoxide ? alkene reaction. ...
-
One of the biological pathways by which an amine is converted to a ketone involves two steps: (1) oxidation of the amine by NAD+ to give an imine, and (2) hydrolysis of the imine to give a ketone...
-
In Problems 712, determine whether the given sequence is arithmetic, geometric, or neither. If the sequence is arithmetic, find the common difference and the sum of the first n terms. If the sequence...
-
Colin Smith was formerly a corporal in the British Army and saw active service in Afghanistan. One day on patrol he was shot in the leg by a sniper and was lying on the ground, bleeding heavily. His...
-
Submit a comprehensive bi-weekly report documenting noteworthy occurrences and pivotal decisions undertaken within the Investment Game Project. This report should emphasize analyses grounded in risk...
-
For this assignment, we need to research and compare international and U.S. accounting standards. This will enable you to see how the different reporting methods affect business and how product costs...
-
c) Give the example of two matrices A and B of order 2 x 2 such that AB is not equal to BA. d) Find the adjoint of the matrix A = [23]
-
This question is related to Stocks valuation in Financial Management subject. We calculated share price of ABC company as 4.58$ with the help of Discounted Free Cashflow method. When used P/E ratio...
-
There are 25 students in Mrs. Bushs sixth-grade class. On a cold winter day in February, many of the students had runny noses and sore throats. After examining each student, the school nurse...
-
In the operation of an automated production line with storage buffers, what does it mean if a buffer is nearly always empty or nearly always full?
-
Use data from Table 10.3 to estimate the enthalpy change ( r H) for the following reaction. Table 10.3 CH6(g) + Cl2(g) CH5Cl(g) + HCl(g) AH = ?
-
Show how you might prepare aniline from each of the following compounds: (a) Benzene (b) Bromobenzene (c) Benzamide
-
Show how you might synthesize each of the following compounds from 1-butanol: (a) Butylamine (free of 28 and 38 amines) (b) Pentylamine (c) Propylamine (d) Butylmethylamine
-
Show how you might convert aniline into each of the following compounds. (You need not repeat steps carried out in earlier parts of this problem.) (a) Acetanilide (b) N-Phenylphthalimide (c)...
-
3. Describe the stages of anaesthesia based on increasing depth of CNS depression. (4 Marks) 4. Describe three advantages and three disadvantages of local anesthesia. (6 marks)
-
calculcate AI, Zi, Av, Ap & Zo for the CE tramsistor amplifier. Laving source inter- mel resistance 800-2 & terminated with 9 load Resistance of 2 kr Ret the transistor parameters have:- Rs = 800-2,...
-
Using the balance sheet for DEF Enterprises as of December 31, 2023, assess the companys financial leverage and stability. DEF Enterprises Balance Sheet As of December 31, 2023 Assets Current Assets...

Study smarter with the SolutionInn App


