(a) For the solidification of iron, calculate the critical radius r* and the activation free energy ?G*...
Question:
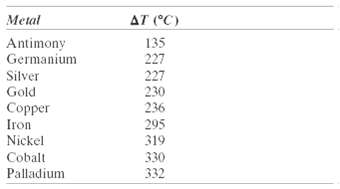
(a) For the solidification of iron, calculate the critical radius r* and the activation free energy ?G* if nucleation is homogeneous. Values for the latent heat of fusion and surface free energy are ???1.85 ?? 109 J/m3 and 0.204 J/m2, respectively. Use the supercooling value found in Table 10.1.
(b) Now calculate the number of atoms found in a nucleus of critical size. Assume a lattice parameter of 0.292 nm for solid iron at its melting temperature.
Transcribed Image Text:
Metal AT (°C) Antimony Germanium 135 227 227 230 236 Silver Gold Copper Iron Nickel 295 319 Cobalt 330 Palladium 332
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (19 reviews)
a This portion of the problem asks that we compute r and G for the homogeneous nucleation of the sol...View the full answer

Answered By

Nicholas Maina
Throughout my tutoring journey, I've amassed a wealth of hands-on experience and honed a diverse set of skills that enable me to guide students towards mastering complex subjects. My proficiency as a tutor rests on several key pillars:
1. Subject Mastery:
With a comprehensive understanding of a wide range of subjects spanning mathematics, science, humanities, and more, I can adeptly explain intricate concepts and break them down into digestible chunks. My proficiency extends to offering real-world applications, ensuring students grasp the practical relevance of their studies.
2. Individualized Guidance:
Recognizing that every student learns differently, I tailor my approach to accommodate various learning styles and paces. Through personalized interactions, I identify a student's strengths and areas for improvement, allowing me to craft targeted lessons that foster a deeper understanding of the material.
3. Problem-Solving Facilitation:
I excel in guiding students through problem-solving processes and encouraging critical thinking and analytical skills. By walking learners through step-by-step solutions and addressing their questions in a coherent manner, I empower them to approach challenges with confidence.
4. Effective Communication:
My tutoring proficiency is founded on clear and concise communication. I have the ability to convey complex ideas in an accessible manner, fostering a strong student-tutor rapport that encourages open dialogue and fruitful discussions.
5. Adaptability and Patience:
Tutoring is a dynamic process, and I have cultivated adaptability and patience to cater to evolving learning needs. I remain patient through difficulties, adjusting my teaching methods as necessary to ensure that students overcome obstacles and achieve their goals.
6. Interactive Learning:
Interactive learning lies at the heart of my approach. By engaging students in discussions, brainstorming sessions, and interactive exercises, I foster a stimulating learning environment that encourages active participation and long-term retention.
7. Continuous Improvement:
My dedication to being an effective tutor is a journey of continuous improvement. I regularly seek feedback and stay updated on educational methodologies, integrating new insights to refine my tutoring techniques and provide an even more enriching learning experience.
In essence, my hands-on experience as a tutor equips me with the tools to facilitate comprehensive understanding, critical thinking, and academic success. I am committed to helping students realize their full potential and fostering a passion for lifelong learning.
4.90+
5+ Reviews
16+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
(a) For the solidification of nickel, calculate the critical radius r* and the activation free energy G* if nucleation is homogeneous. Values for the latent heat of fusion and surface free energy are...
-
The critical values of r in Table A-6 are found by solving for r to get where the t value is found from Table A-3 by assuming a two-tailed case with n - 2 degrees of freedom. Table A-6 lists the...
-
The lattice energy of an ionic solid such as NaCl is the enthalpy change H° for the process in which the solid changes to ions. For example, NaCl(s) Na+(g) + Cl(g) H = 786 kJ/mol Assume that the...
-
Jansen, Inc., is a defense contractor that uses job costing. Because the firm uses a perpetual inventory system, the three supporting schedules to the income statement (the schedule of raw materials...
-
It had been a very bad morning for John Ross, the general manager of MMC's Chinese joint venture. He had just gotten off the phone with his boss in St Louis, Phil Smith, who was demanding to know why...
-
During 2020, Global Designs Inc. had the following transactions. Aug. 31 Traded in furniture with a cost of $42,000 and accumulated depreciation of $25,800 recorded in the accounting records on this...
-
What is a merchants firm offer?
-
Revenue recognition at and after time of sale. Assume that during December 2008, Nordstrom sold $20 million of merchandise and another $12 million of gift cards, of which $24 million was on credit...
-
A manufacturing firm reported the following sales and operating data for its 3 segments: Sales Average Operating Assets Net Operating Income Minimum Required Rate of Return Segment 1 Segment 2...
-
Skunk Hollow, Arkansas, is proud of their energy conservation program. They want to know how much energy they have saved. A sample of 40 houses reveals an average saving of 47.5 kilowatt hours per...
-
If copper (which has a melting point of 1085C) homogeneously nucleates at 849C, calculate the critical radius given values of 1.77 109 J/m3 and 0.200 J/m2, respectively, for the latent heat of...
-
(a) Assume for the solidification of iron (Problem 10.4) that nucleation is homogeneous, and the number of stable nuclei is 106 nuclei per cubic meter. Calculate the critical radius and the number of...
-
As this chapter is about self-management, what stressors do you anticipate, and how do you plan to manage your stress response?
-
1. Gluteus maximus muscle and mention the structures covered by it.
-
2. External features and relations of Kidney.
-
3. Features of Synovial joint and its classification.
-
1. Name the paranasal air sinuses.
-
2. Name the ventricles of Brain.
-
What are some important factors in deciding whether to use equity or cash in an acquisition?
-
After Theorem 1.5 we note that multiplying a row by 0 is not allowed because that could change a solution set. Give an example of a system with solution set S0 where after multiplying a row by 0 the...
-
List reasons why there might be a connection between the amount of energy used by a country and its economic development.
-
The microstructure of an iron-carbon alloy consists of proeutectoid cementite and pearlite; the mass fractions of these micro constituents are 0.11 and 0.89, respectively. Determine the concentration...
-
Given here are the solidus and liquids temperatures for the copper-gold system. Construct the phase diagram for this system and label each region. Composition (wt% Au) Solidus Temperature (C)...
-
Consider 1.5 kg of a 99.7 wt% Fe-0.3 wt% C alloy that is cooled to a temperature just below the eutectoid. (a) How many kilograms of proeutectoid ferrite form? (b) How many kilograms of eutectoid...
-
Callaho Inc. began operations on January 1 , 2 0 1 8 . Its adjusted trial balance at December 3 1 , 2 0 1 9 and 2 0 2 0 is shown below. Other information regarding Callaho Inc. and its activities...
-
Required: 1. Complete the following: a. Colnpute the unit product cost under absorption costing. b. What is the company's absorption costing net operating income (loss) for the quarter? c. Reconcile...
-
Bond Valuation with Semiannual Payments Renfro Rentals has issued bonds that have an 8% coupon rate, payable semiannually. The bonds mature in 6 years, have a face value of $1,000, and a yield to...

Study smarter with the SolutionInn App


