An equimolar solution of benzene and toluene is totally evaporated at a constant temperature of 90C. What
Question:
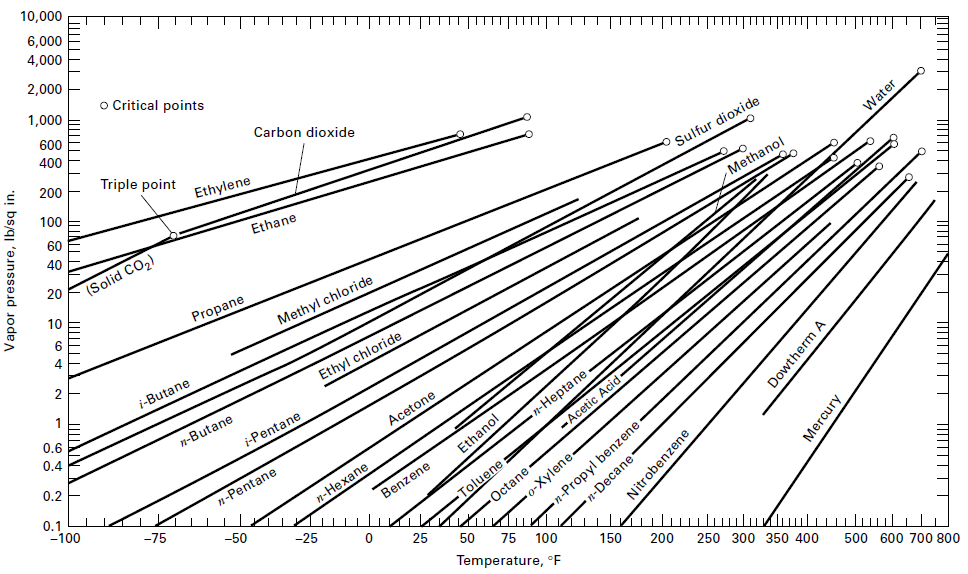
An equimolar solution of benzene and toluene is totally evaporated at a constant temperature of 90°C. What are the pressures at the beginning and end of the vaporization process? Assume an ideal solution and use the vapor pressure curves ofFigure.
Transcribed Image Text:
10,000 6,000 4,000 2,000 П| o Critical points 1,000 600 400 Carbon dioxide 200 Triple point TП 100 Ethylene 60 40 Ethane Sulfur dioxide Water 20 (Solid CO2) Methanol 10 Propane 4 Methyl chloride i-Butane Ethyl chloride 0.6 0.4 n-Butane i-Pentane 0.2 Acetone n-Hepta ne 0.1 -100 -Acețic Acid- n-Pentane Ethanol Octane -o-Xylene- n-Propyl benzene п-Нехапe -75 KIn ulu Toluene: Benzene -50 -25 25 75 100 Temperature, °E 150 200 250 300 350 400 500 600 700 800 Vapor pressure, Ib/sqir ШІ n-Decane Nitrobenzene Dowtherm A Mercury
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Substitution of Raoults law Eq 3 in Table 23 ...View the full answer

Answered By

Sinmon Warui Kamau
After moving up and down looking for a job, a friend introduced me to freelance writing. I started with content writing and later navigated to academic writing. I love writing because apart from making a living out of it, it is also a method of learning and helping others to learn.
5.00+
40+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Chemical Engineering questions
-
A liquid mixture of benzene and toluene is to be separated in a continuous single-stage equilibrium flash tank. The pressure in the unit may be adjusted to any desired value, and the heat input may...
-
A liquid mixture of benzene and toluene is to be separated in a continuous single-stage equilibrium flash tank. The pressure in the unit may be adjusted to any desired value, and the heat input may...
-
A vertical flat plate is maintained at a constant temperature of 120oF and exposed to atmospheric air at 70oF. At a distance of 14 in. from the leading edge of the plate the boundary layer thickness...
-
Under what circumstances should derived associations be used?
-
Refer to E8-10. Deere & Company reports in the notes to its financial statements that its trade accounts and notes receivable have significant concentrations of credit risk in the agricultural,...
-
From the following, calculate the net cash flows from operating activities (use the direct method): Sales $9,300 Cost of Goods Sold 4,400 Salaries Expense 1,400 Insurance Expense 840 Other Expenses...
-
Someone uses the PewHealth dataset to run a regression model that shows that various types of non-married people have different average scores on the Cell-phone Use Index. Someone else looks at this...
-
On June 15, Palmyra Company sold merchandise for $5,200 on terms of n/30 to Lim Company. On June 20, Lim Company returned some of the merchandise for a credit of $1,200, and on June 25, Lim paid the...
-
Ship Fast Corporation is an overnight shipper. A (Click the icon to view additional information.) At year-end, Ship Fast ended with the foregoing May 31, 2019, balances. Read the requirements...
-
Use the following narrative to complete the UML class diagram with classes, associations, and multiplicities outlined below and then answer the associated questions: The Nordic Construction Companys...
-
The following mixture is introduced into a distillation column as saturated liquid at 1.72MPa. Calculate the bubble-point temperature using the K-values of Figure. Compound kmol/h Ethane 1.5 Propane...
-
The following equations are given for the liquid-phase activity coefficients of the water (W)-acetic acid (A) system. Find the dew point and bubble point of a mixture of composition xw = 0.5, xA =...
-
List five distinct types of financial intermediaries, explain what they do, and provide a comparison/contrast of the basic intermediation services they provide.
-
Your task is to check the internet and the?Common Vulnerabilities and Exposures (CVE) List?for networked IoT or?IoMT?devices with publicly known problems identified in the past six months.?? Select...
-
The first quarter tax return needs to be filed for Prevosti Farms and Sugarhouse by April 15, 2021. For the taxes, assume the second February payroll amounts were duplicated for the March 5 and March...
-
ABC Boating had a large fire that destroyed many of their boats ready to go to market. ABC decided to lay off a large number of employees rather than continue to pay them. The wages that this group...
-
HOW BEER IS MADE The beer-making process is an art. It takes time, patience and quite a bit of experimentation to find that perfect flavour. Once you have found the right mix, you can replicate it,...
-
Justine, an underwriter at NewWorld Insurance is working with her company's premium auditing department. Which one of the following is true regarding this collaboration? Available answer options...
-
E-Signatures. Stewart Lamle invented Farook, a board game similar to Tic Tac Toe. In May 1996, Lamle began negotiating with Mattel, Inc., to license Farook for distribution outside the United States....
-
What services are provided by the provincial and territorial governments?
-
Define the Sortino and Omega ratios and discuss their value in assessing ETF performance.
-
It is required to absorb 96% of the benzene from a gas stream with absorption oil in a sieve-tray column at a nominal pressure of 1 atm. The feed conditions are as follows: Tray geometry is as...
-
A ternary mixture of methanol, ethanol, and water is distilled in a sieve-tray column to obtain a distillate with not more than 0.01 mol% water. The feed to the column is as follows: Flow rate,...
-
A bubble-cap tray absorber is designed to absorb 40% of the propane from a rich gas at 4 atm. The specifications for the entering rich gas and absorbent oil are as follows: (a) Determine the number...
-
DETAILS 1. [-/1 Points) SMITHNM13 11.2.025. MY NOTES Convert the credit card rate to the APR. Oregon, 2% per month % Need Help? ReadIt Watch
-
Corom Stack Standard CALCULATOR PRINTER VERSION BACK NEXT Problem 13-02A a-c (Part Level Submission) Sheffield Corporation had the following stockholders' equity accounts on January 1, 2020: Common...
-
Suppose that you own 2,100 shares of Nocash Corp. and the company is about to pay a 25% stock dividend. The stock currently sells at $115 per share. a. What will be the number of shares that you hold...

Study smarter with the SolutionInn App


