Assume that you need to prepare 5-cholesten-3-one from cholesterol. How could you use IR spectroscopy to tell
Question:
Assume that you need to prepare 5-cholesten-3-one from cholesterol. How could you use IR spectroscopy to tell whether the reaction was successful? What differences would you look for in the IR spectra of starting material and product?
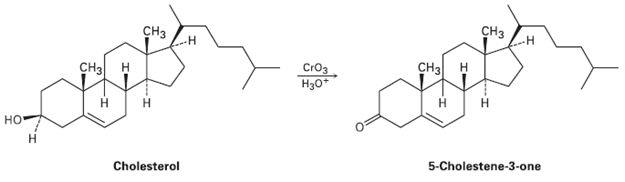
Transcribed Image Text:
ҫHз СHз Сн CHз н CHа н CrOз H30* н но Н 5-Cholestene-3-one Cholesterol -т
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
The infrared spectra of cholesterol and 5cholestene3one each exhibit a unique absorp...View the full answer

Answered By

Gilbert Chesire
I am a diligent writer who understands the writing conventions used in the industry and with the expertise to produce high quality papers at all times. I love to write plagiarism free work with which the grammar flows perfectly. I write both academics and articles with a lot of enthusiasm. I am always determined to put the interests of my customers before mine so as to build a cohesive environment where we can benefit from each other. I value all my clients and I pay them back by delivering the quality of work they yearn to get.
4.80+
14+ Reviews
49+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Assume that you need to prepare 4-methyl-2-pentyne and discover that the only alkynes on hand are acetylene and propyne. You also have available methyl iodide, isopropyl bromide, and 1,...
-
How could you use IR spectroscopy to distinguish between the following pairs of isomers? a. b. c. (CH3CH2)3N and (CH3CH2CH2)2NH 0 CH.CCH,CH, and CH CHCH-CH, CH3 CHCHO and CH CHOCH
-
What features would you look for in a social media outlet? What types of information would you avoid making part of your social media strategy?
-
In September 2020 Kevin sells a drawing for 2,000. He bought the drawing in February 2014 for 50,000 when it was thought (incorrectly) to be by a famous artist. Compute the allowable loss.
-
What are three types of opportunities for sharing that form a sound basis for diversification or vertical integration? Give an example of each from companies you have read about.
-
Knight Fashion store operates three departments: Mens, Womens, and Accessories. Knight Fashion allocates all fixed expenses (unavoidable building depreciation and utilities) based on each departments...
-
What was the average annual return during this period? a. -4.40% b. 3.10% c. 8.69% d. 12.19%
-
Discuss why organizational design and communication flow are so closely related.
-
Larkspur, Inc. has the following income statement (in millions): LARKSPUR, INC. Income Statement For the Year Ended December 31, 2023 Net Sales $300 Cost of Goods Sold 160 Gross Profit 140 Operating...
-
The comparative statements of Larker Tool Company are presented below. Larker Tool Company Income Statement For the Years Ended December 31 Larker Tool Company Statements of Financial Position...
-
Show the mechanism of the reaction of p-methaylphenol with 2-methylpropene and H3PO4 catalyst to yield the food additive BHT.
-
When the 1HNMR spectrum of an alcohol is run in dimethyl sulfoxide (DMSO) solvent rather than in chloroform, exchange of the OH proton is slow and spinspin splitting is seen between the OH proton...
-
In Problem solve the equation for the unknown quantity. 6.75 = 3(1 + i) 2
-
Die sequence (5) As Integer sequence (1) 2 sequence(0) 10 For i As Integer sequence (1) Next 2 To sequence.Length - 1 sequence(12) sequence(i-1) Write ONLY 1 line of code that declares the array and...
-
2. Convert the following hexadecimal numbers into decimal (5 points each): a. OB b. F1 C. 11 d. H5 e. 42
-
Required 1. By looking at the financial statements provided would you say the management is doing a 'good job' in managing the company ? justify your answer 2. Would you invest in this Company after...
-
Diversity has been a buzzword in organizational behavior for a long time.Recently, terms like inclusion and cultural sensitivity have been more frequently used.Currently, the term most often...
-
As you watch the movie: "The environmental tourist," take note of the different destinations being highlighted. What are some of the issues in the safari parks in Africa? What initiatives are...
-
What are the effects on a report on reportable conditions when the auditor separately identifies and describes material weaknesses?
-
A seasonal index may be less than one, equal to one, or greater than one. Explain what each of these values would mean.
-
The following data are given for CCl 4 . Normal melting point, -23 C; normal boiling point, 77 C; density of liquid 1.59 g/mL; fus H = 3.28 kJ mol -1 ; vapor pressure at 25 C, 110 Torr. (a) What...
-
(a) What is the index of hydrogen deficiency of 2-hexene? (b) Of methylcyclopentane? (c) Does the index of hydrogen deficiency reveal anything about the location of the double bond in the chain? (d)...
-
Zingiberene, a fragrant compound isolated from ginger, has the molecular formula C15H24 and is known not to contain any triple bonds. (a) What is the index of hydrogen deficiency of zingiberene? (b)...
-
Carbonyl groups also count for a unit of hydrogen deficiency. What are the indices of hydrogen deficiency for the reactant and for the product in the equation shown at the beginning of Section 4.16...
-
31 BT E R ole]1i 1o 2111Trial balancev For the Quarter Ended May 31, 2022For the Year Ended May 31, 2022May 31, 2022 Totals $ $ Current Attempt in Progress Julia Dumars is a licensed CPA. During the...
-
Chief Customer Officer Jayden Hayden has asked sales analyst Paige Sage to analyze the company's Western Kentucky district. Paige intends to calculate ROAM for the district. She has compiled the...
-
Which of the following statements is correct?If the demand curve for a firm's stock is relatively flat, the firm will have a more difficult time raising alarge amount of new equity funds for...

Study smarter with the SolutionInn App


