At what position would you expect electrophilic substitution to occur in each of the followingsubstances? (a) (b)
Question:
At what position would you expect electrophilic substitution to occur in each of the followingsubstances?
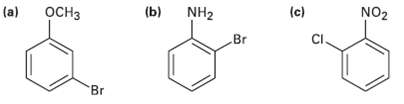
Transcribed Image Text:
(a) (b) (c) оснз NH2 Br NO2 CI. Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
a OCH3 OCH 3 OCH3 E Br Br Br E Both groups are orthopara directors and direct substitution ...View the full answer

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
At what position would you expect to observe IR absorptions for the following molecules?
-
In each of the following instances, would you expect a positive, negative, or zero correlation? (a) Number of salespersons and total dollar sales for real estate firms. (b) Total payroll and percent...
-
What can you say about the series Σ an in each of the following cases? (b) lim an (a) lim 0.8 an+1 (c) lim
-
The number of claims filed each week with Security Insurance Company has a mean of 700 and a standard deviation of 250. Calculate the probability that the number of claims this week will be: (a)...
-
In 2016, Geoff incurred $900,000 of mine and exploration expenditures. He elects to deduct the expenditures as quickly as the tax law allows for regular income tax purposes. a. How will Geoff's...
-
Labrie Hospital wants to analyze its revenue per patient day. A patient day is one patient staying for one day. The following information has been provided to you: Required A. Determine the revenue...
-
What are at least five strategies followed by smart shoppers.
-
The following items were discovered during the December 31, 2011 audit of the financial statements of Westmoreland Corporation: 1. The company's financial statements did not include an accrual for...
-
A CPA is auditing financial statements of a non-issuer. The CPA will state that evidence has been examined on a test basis in which section of the CPA's report? a. Responsibilities of Management b....
-
Jeter Co. uses a perpetual inventory system and both an accounts receivable and an accounts payable subsidiary ledger. Balances related to both the general ledger and the subsidiary ledger for Jeter...
-
Draw resonance structures for the intermediates from reaction of an electrophile at the ortho, Meta and Para positions of nitrobenzene. Which intermediates are most stable?
-
Show the major product(s) from reaction of the following substances with (i) CH3CH2Cl, AlCl3 and (ii) HNO3,H2SO4 (b) (a)
-
What opportunities and potential problems are posed by the formation of more than one coalition within an organization, each one advocating a different direction or alternatives? What steps can you...
-
Below is the amount of widgets that Adam, Robert, and Karl are each willing to make at three different prices. Producer $1 per widget $2 per widget $3 per widget Adam 3 Robert 5 Karl 3 6 7 9 a. What...
-
A depreciable asset with a cost of $40550 has a residual value of $3000 and a useful life of 5 years. Total estimated units of output are 75100 and in year 1 a total of 5400 units were produced....
-
How many kilograms of lead must be added to 71 kg oftin to yield a Pb-Sn alloy having a composition of 71 at%Sn-29 at% Pb? mpb= _____ kg
-
Kayla consumes about 100 grams of protein each day. Of this, 25 grams is broken down and used to make ATP, and 25 grams is used to synthesize (make) body fat. What does this tell you about Kayla's...
-
During the year, Park Corporation had 100.000 shares of $10 par value common stock and 20,000 shares of 89. $50 par value convertible preferred stock outstanding. Each share of preferred stock may be...
-
What are the effects on the auditor's standard report of the decision to (a) make reference and (b) not make reference to another auditor?
-
A 6-lb shell moving with a velocity ?? v0k explodes at point D into three fragments which hit the vertical wall at the points indicated. Fragments A, B, and C hit the wall 0.010 s, 0.018 s, and 0.012...
-
Derive the equations necessary to expand Illustration 6.4-1 to include the thermodynamic state variables internal energy, Gibbs energy, and Helmholtz energy. Illustration 6.4-1 Making of a...
-
Show the major products of these eliminationreactions: CI + NaOCH,CH3 Ph + NaOCH,CH, ELOH ELOH a) Ph Br d) Ph + NaOH CH;OH c) N(CH3)3 OTs
-
When 2-methyl-2-propanol is treated with sulfuric acid, 2-methylpropene is formed. Show all of the steps in the mechanism for this reaction. Don't forget to use curved arrows to show the movement of...
-
Show all of the steps in the mechanism for this reaction, what other products would you expect to beformed? CH3 Br CH3 . CH,O -CH,CH CH.C- C=C CH,CH3 CH3 .
-
Whenever the slope of the total product curve is increasing at an increasing rate, the marginal product curve is _ _ _ _ _ . a . positive and rising b . positive and falling c . negative and falling...
-
When a Pigouvian tax is imposed, _ _ _ _ _ _ _ _ . A ) the marginal social benefit curve shifts downward B ) the marginal private cost curve shifts upward C ) the marginal social cost curve shifts...
-
Match the term to the phrase: Group of answer choices The macroeconomic model that uses aggregate demand and aggregate supply to determine and explain the price level and level of real domestic...

Study smarter with the SolutionInn App


