Calculate the number density of free electrons for (a) Mg (? = 1.74 g/cm3) and (b) Zn
Question:
Calculate the number density of free electrons for
(a) Mg (? = 1.74 g/cm3) and
(b) Zn (? = 7.1 g/cm3), assuming two free electrons per atom, and compare your results with the values listed in Table 27-1.
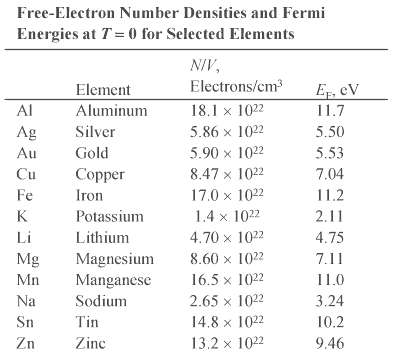
Transcribed Image Text:
Free-Electron Number Densities and Fermi Energies at T= 0 for Selected Elements NIV, Electrons/cm E, cV Element Al Aluminum 18.1 x 1022 11.7 Silver 5.50 Ag 5.86 x 1022 5.53 Au Gold 5.90 x 1022 7.04 Cu Copper 8.47 x 1022 Fe Iron 17.0 x 1022 11.2 K Potassium 1.4 x 1022 2.11 Li Lithium 4.70 x 1022 4.75 8.60 x 1022 Mg Magnesium Manganese 7.11 Mn 16.5 x 1022 11.0 Na Sodium 2.65 x 1022 3.24 14.8 x 1022 Sn Tin 10.2 Zn Zinc 13.2 x 1022 9.46
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 84% (13 reviews)
a b n 2 N A M a n 2 602 10 23 174243 n 862 10 22 b n 1...View the full answer

Answered By

Irfan Ali
I have a first class Accounting and Finance degree from a top university in the World. With 5+ years experience which spans mainly from the not for profit sector, I also have vast experience in preparing a full set of accounts for start-ups and small and medium-sized businesses. My name is Irfan Ali and I am seeking a wide range of opportunities ranging from bookkeeping, tax planning, business analysis, Content Writing, Statistic, Research Writing, financial accounting, and reporting.
4.70+
249+ Reviews
530+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Electricity and Magnetism questions
-
Calculate the number density of free electrons in (a) Ag (? = 10.5 g/cm3) and (b) Au (? = 19.3 g/cm3), assuming one free electron per atom, and compare your results with the values listed in Table...
-
The number density of free electrons in copper is 8.47 ?? 1022 electrons per cubic centimeter. If the metal strip in Figure is copper and the current is 10 A, find (a) The drift velocity vd and (b)...
-
Compare your results with those obtained in Problem 8.113 (p. 330)? Microbiology Refer to the data in Table 8.28 (p. 330). Table 8.28 Pod weight (g) from inoculated (I) and uninoculated (U) plantsa
-
When developing a survey instrument for a cross-country study, market researchers often need to construct a scale (e. g., a 7-point disagree/agree scale). What are the major items that one should be...
-
In 2015, CSX Corporation, which operates under the name Surface Transportation, reported operating expenses of $8,227 million. A partial list of the company's operating expenses follows. CSX...
-
The Sherill Utility District was recently established. Its balance sheet, after one year, is presented below. Note the following additional information: ¢ The general fund received all of its...
-
Service Revenue} Melrose Milk Delivery provides weekly gourmet milk delivery to the residents of Nicetown. Melrose charges each customer \(\$ 45\) per week for its milk delivery, and it received...
-
Thomsonetics, Inc., a rapidly growing early stage technology company, had the pretax income noted below for calendar years 2010-2012. The firm was subject to corporate taxes consistent with the rates...
-
There is a % column right beside each balance sheet account for each year. Go back and look at the template provided. The text explains the difference between a balance sheet and a common - size...
-
Honeybee Hippie is a retail store specializing in womens clothing. The store has established a liberal return policy for the holiday season in order to encourage gift purchases. Any item purchased...
-
How does the change in the resistivity of copper compare with that of silicon when the temperature increases?
-
Estimate the fraction of free electrons in copper that are in excited states above the Fermi energy at (a) Room temperature of 300 K and (b) 1000 K.
-
New Wave Pool Corporation is authorized to issue common and $3 convertible preferred shares. Each preferred share is convertible into four common shares. On July 2, the company issued 100,000...
-
On January 1, 2018, Trueblood, Inc. purchased a piece of machinery for use in operations. The total acquisition cost was $33,000. The machine has an estimated useful life of three years and a...
-
Robert is designing a new Active Directory Domain Services infrastructure for a company called Litware, Inc., which has its headquarters in New York and two additional offices in London and Tokyo....
-
Lycan is a functionalist , which means that he believes that if something functions like a person, then it is a person . For Lycan, function is more important than origin. N.B. human = a particular...
-
202380-Fall 2023-ACCT-3324 X M Question 3 - Chapter 14 - Integ x C F26 McGraw Hill Excel Question - Saved File Home Insert Draw Formulas Data Review View Help 5v A v...
-
Financial data for Bridger Inc. for last year are as follows: BRIDGER INC. Balance Sheet Ending Balance Beginning Balance Assets Cash $ 165,000 $ 160,000 Accounts receivable 390,000 250,000 Inventory...
-
Nygren Supply Company uses a voucher register similar to the one described in this chapter. Instructions: Journalize the transactions completed during February of the current year. Use page 2 of a...
-
Audrey purchases a riding lawnmower using a 2-year, no-interest deferred payment plan at Lawn Depot for x dollars. There was a down payment of d dollars and a monthly payment of m dollars. Express...
-
At 25C, what is the mass in grams of 1000.0 mL of water?
-
What is the maximum speed with which a 1050-kg car can round a turn of radius 77m on a flat road if the coefficient of static friction between tires and road is 0.80? Is this result independent of...
-
Calculate the speed of a satellite movie in a stable circular orbit about the Earth at a height of 3600 km.
-
How large must the coefficient of static friction be between the tires and the road if a car is to round a level curve of radius 85m at a speed of 95km/h?
-
Lovell purchased a used van for use in its business on January 1 , 2 0 2 0 . It paid $ 1 7 , 0 0 0 for the van. Lovell expects the van to have a useful life of four years, with an estimated residual...
-
Sheridan Corporation had the following 2 0 2 5 income statement. \ table [ [ Sales revenue, ] , [ Cost of goods sold,$ 2 2 0 , 0 0 0
-
Sheridan Company issues $ 5 0 8 0 0 0 0 , 6 . 0 % , 5 - year bonds dated January 1 , 2 0 2 4 on January 1 , 2 0 2 4 . The bonds pay interest semiannually on June 3 0 and December 3 1 . The bonds are...

Study smarter with the SolutionInn App


