Calculate the reduced mass for the H35Cl and H37Cl molecules and the fractional difference ??/. Show that
Question:
Calculate the reduced mass for the H35Cl and H37Cl molecules and the fractional difference ??μ/μ. Show that the mixture of isotopes in HCl leads to a fractional difference in the frequency of a transition from one rotational state to another given by ??f/f = ??μ/μ. Compute ??f/f and compare your result withFigure.
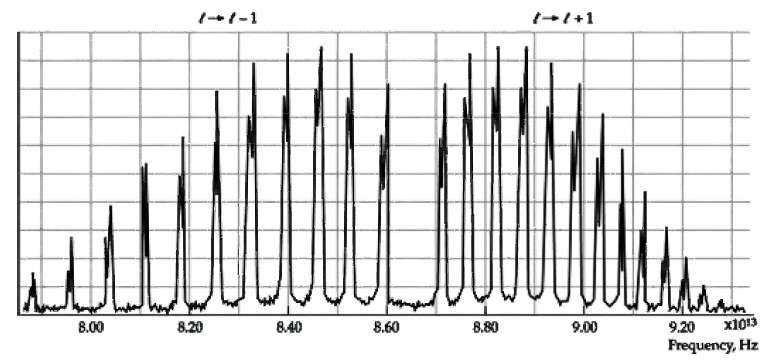
Transcribed Image Text:
1+1-1 x1013 8.20 8.40 B.00 8.60 8.80 9.00 9.20 Frequency, Hz
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
We shall express the results in unified mass units For ...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Modern Physics questions
-
Show that a mixture of saturated liquid water and saturated water vapor at 100C satisfies the criterion for phase equilibrium.
-
Show that a mixture of saturated liquid water and saturated water vapor at 300 kPa satisfies the criterion for phase equilibrium.
-
Show that a mixture of saturated liquid water and saturated water vapor at 300 kPa satisfies the criterion for phase equilibrium.
-
Various streptococci and lactobacilli were traditionally grouped together as lactic acid bacteria because of their characteristic fermentation. Most of them were found to have a DNA Guanine plus...
-
Record the following transactions at (a) gross and (b) net: 201X Sept. 3 Bought merchandise on account from Curik Co.; terms 3/10, n/30, $7,500. Voucher no. 32 was prepared. 18 Issued check no. 479...
-
How is investing related to liquidity? Give some examples of liquid investments.
-
Have you come across examples of American formalisation, Japanese centralisation and European socialisation? How do they conflict? What is the practical implication of these variations? LO1
-
Use Appendix B.5 to locate the value of t under the following conditions. a. The sample size is 15 and the level of confidence is 95%. b. The sample size is 24 and the level of confidence is 98%. c....
-
Refer to the transactions listed in the case study below Capture the transactions in the correct joumals, also known as books of first entry. Remember to take VAT into consideration. Transactions 1 ....
-
Two partners have decided to acquire a more expensive manufacturing facility. One of the partners researched and found a factory selling for $25 million. The partners went to a local bank to discuss...
-
Two objects of mass m 1 and m 2 are attached to a spring of force constant K and equilibrium length r 0 . (a) Show that when m 1 is moved a distance ?r 1 from the center of mass, the force exerted by...
-
In calculating the rotational energy levels of a diatomic molecule, we did not consider rotation of the molecule about the line joining the atoms. (a) Estimate the moment of inertia of the H2...
-
In Exercises the random variable n represents the number of units of a product sold per day in a store. The probability distribution of n is given by P(n). Find the probability that two units are...
-
James A. and Ella R. Polk, ages 70 and 65, respectively, are retired physicians who live at 3319 Taylorcrest Street, Houston, Texas 77079. Their three adult children (Benjamin Polk, Michael Polk, and...
-
I need help solving the following question: - Thank you in advance. On January 1, Year 6, HD Lid., a building supply company, JC Lid., a construction company, and Mr. Saeid, a private investor,...
-
Let X 1 , , X n X 1 , , X n be a random sample from a normal distribution with mean and variance 1. Find the uniformly minimum variance unbiased estimator of 2 2 .2 answers
-
The ledger of Duggan Rental Agency on March 31 of the current year includes the following selected accounts before adjusting entries have been prepared. Debit Credit Prepaid Insurance $3,600 Supplies...
-
1.Using the Excel file Sales transaction find the following15 Marks a.Identify the levels of measurement for each variables b.Construct a cross tabulation to find the number of transactions by...
-
Southeastern Pennsylvania Transportation Authority [SEPTA] is a regional mass transit authority in the Philadelphia area. SEPTA sought to upgrade the fitness level of its transit police by adopting a...
-
Drainee purchases direct materials each month. Its payment history shows that 65% is paid in the month of purchase with the remaining balance paid the month after purchase. Prepare a cash payment...
-
(a) Find i s ,v 1 ,v 2 , and i l in Fig. 5-18(a). (b) Compare these results with those obtained when source and load are connected directly as in Fig. 5-18(b). R voltage source 11, R voltage source...
-
A certain electronic device needs to be protected against sudden surges in current. In particular, after the power is turned on the current should rise no more than 7.5mA in the first 120s. The...
-
A 25-turn 12.5 cm-diameter coil is placed between the pole pieces of an electromagnet. When the magnet is turned on, the flux through the coil changes, inducing and emf at what rate (in T/s) must the...
-
Calculate the peak output voltage of a simple generator whose square armature windings are 660 cm on a side the armature contains 155 loops and rotates in a field of 0.200T at a rate of 120 rev/s.
-
DETAILS 1. [-/1 Points) SMITHNM13 11.2.025. MY NOTES Convert the credit card rate to the APR. Oregon, 2% per month % Need Help? ReadIt Watch
-
Corom Stack Standard CALCULATOR PRINTER VERSION BACK NEXT Problem 13-02A a-c (Part Level Submission) Sheffield Corporation had the following stockholders' equity accounts on January 1, 2020: Common...
-
Suppose that you own 2,100 shares of Nocash Corp. and the company is about to pay a 25% stock dividend. The stock currently sells at $115 per share. a. What will be the number of shares that you hold...

Study smarter with the SolutionInn App


