Epoxide?s react with Grignard reagents to yield alcohols propose a mechanism. 1. CHMgBr 2. *
Question:
Epoxide?s react with Grignard reagents to yield alcohols propose a mechanism.
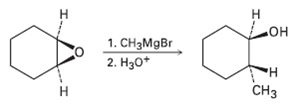
Transcribed Image Text:
Н н 1. CHзMgBr 2. Нао* но° н CHз Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
I 0 H CH3 H3O FCH3 SN2 reaction ...View the full answer

Answered By

Usman Nasir
I did Master of Commerce in year 2009 and completed ACCA (Association of Chartered Certified Accountants) in year 2013. I have 10 years of practical experience inclusive of teaching and industry. Currently i am working in a multinational company as finance manager and serving as part time teacher in a university. I have been doing tutoring via many sites. I am very strong at solving numerical / theoretical scenario-based questions.
4.60+
16+ Reviews
28+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
(a) Unlike other esters, which react with Grignard reagents to give tertiary alcohols, ethyl Formate yields a different class of alcohols on treatment with Grignard reagents. What kind of alcohol is...
-
Nitrites (RC ? N) react with Grignard reagents (R'MgBr). The reaction product from 2-methyipropanenitrile with methyl magnesium bromide has the following spectroscopic properties. Propose a...
-
Grignard reagents react slowly with oxetane to produce primary alcohols. Propose a mechanism for this reaction, and suggest why oxetane reacts with Grignard reagents even though most ethers do not....
-
Use the same information as in question 4, except that Hughes has a debit balance of $5,000 in its Allowance for Doubtful Accounts before any adjustments are made at the end of the year. In this...
-
Explain when and why it is important for a company to globalize.
-
Compare and contrast a cost centre, a revenue centre, a profit centre, and an investment centre. List a specific example of each type of responsibility centre. How is the performance of managers...
-
Your company is evaluating a new project, and you've estimated that the cost of capital for the project will be 8 percent. Assume that you have also projected the following cash flows for the...
-
On January 1, 2013, Gless Textiles issued $12 million of 9%, 10-year convertible bonds at 101. The bonds pay interest on June 30 and December 31. Each $1,000 bond is convertible into 40 shares of...
-
Irbid industrial firm has allowed capacity for selling and production by 1000 units from product (X) and the estimated cost for each unit as the following: JD 120 direct materials, JD 40 variable...
-
Alaska Airlines is unique among the nine major U.S. carriers not only for its extensive flight coverage of remote towns throughout Alaska (it also covers the U.S., Hawaii, and Mexico from its primary...
-
Acid-catalyzed dehydration of 2, 2-dimethylcyclohexanol yields a mixture of 1, 2-dimethylcyclohexene and isopropylidenecyclopentane. Propose a mechanism to account for the formation of bothproducts....
-
How would you prepare the following substances from Cyclopentanol? More than one step may be required. (a) Cyclopentanone (b) Cyclopentane (c) 1-Methylcyclopentanol (d) Trans-2-Methylcyclopentanol I...
-
Critically examine the usefulness or not to marketers of the theory of adoption and diffusion of innovations.
-
You are an Associate Justice of the Supreme Court of Nowhere. The case of Millcreek Corporation u. Simmons, Executor, is now under advisement. Set out below are draft opinions by our colleagues. You...
-
A. This case is being determined by a judge. What type of law is created by judges and decisions madein the courts? B Whatis the name given to a case in law that is a judgement or decision of a court...
-
Simplify the compound fractional expression. x-9x+2 X-10 x+1 *+9
-
The production manager of Rordan Corporation has submitted the following quarterly production forecast for the upcoming 1st Quarter 2nd Quarter 3rd Quarter 9,800 7,900 Units to be produced 7,500 4th...
-
Opening Case When a hospitality and tourism organisation looks for strategic alliance partnerships, it needs to analyse the external and internal environments. Their strategic alliance decisions are...
-
Identify the tests of details that an auditor may use in auditing a payroll bank account.
-
d. The characteristic equation of a control system is given by s+2s+8s+12s+20s+16+16=0. Determine the number of the roots of the equation which lie on the imaginary axis of s-plane
-
Intermolecular forces play vital and varied roles in nature. For example, these forces enable gecko lizards to climb walls and hang upside down from ceilings, seemingly defying gravity....
-
Compound H is optically active and has the molecular formula C6H10. On catalytic hydrogenation H is converted to I (C6H12) and I is optically inactive. Propose structures for H and I.
-
Aspartame is an artificial sweetener. Give the (R, S) designation for each chirality center of aspartame. Aspartame OCH H3N
-
There are four dimethylcyclopropane isomers. (a) Write three-dimensional formulas for these isomers. (b) Which of the isomers are chiral? (c) If a mixture consisting of 1 mol of each of these isomers...
-
What is a good way to improve efficiency and accuracy in the registration process? a. Email audit results to staff members with instructions to improve. Training takes up too much time. b. Scan a...
-
Should Burton pursue both the thermowell machine purchase and the EE acquisition? Why or why not
-
The work sheet: multiple choice eliminates any need for an audit of the financial statements. is often made available to external decision makers. aids in the preparation of financial statements....

Study smarter with the SolutionInn App


