Explain which functional group(s) is present in the compound that has this IRspectrum: 80 60 40 20
Question:
Explain which functional group(s) is present in the compound that has this IRspectrum:
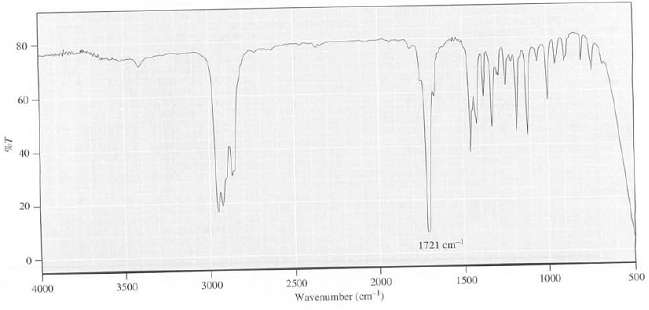
Transcribed Image Text:
80 60 40 20 1721 cm 3500 4000 3000 2500 1500 2000 1000 S00 Wavenimber (cm)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
The intense peak at 1721 cm 1 shows the presence of a CO group The absence ...View the full answer

Answered By

Elias Gichuru
am devoted to my work and dedicated in helping my clients accomplish their goals and objectives,providing the best for all tasks assigned to me as a freelancer,providing high quality work that yields high scores.promise to serve them earnestly and help them achieve their goals.i have the needed expertise,knowledge and experience to handle their tasks.
4.80+
325+ Reviews
859+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
List all types of bonding present in the compound CaCO3. List all types of bonding present in the compound CaCO3. I. ionic bond II. polar covalent bond III. nonpolar covalent bond A. I only B. II...
-
The cesium isotope 137Cs is present in the fallout from aboveground detonations of nuclear bombs. Because it decays with a slow (30.2y) half-life into 137Ba, releasing considerable energy in the...
-
Assume that no censoring is present in the melting times (see the entries in Table 9.1). Estimate S(25), S(30), S(45), and S(55) using both the empirical survival function and the Kaplan-Meier...
-
What is the wavelength of light if its frequency is 1.009 106 Hz?
-
The total number of visitors to an amusement park that stays open all year is given in the table. a. Find a logistic model for the data. b. According to the model, how many potential visitors will...
-
A bag contains 4 white balls and 6 red balls. Three balls are drawn, without replacement, from the bag. (a) What is the probability that all three balls are white? (b) What is the probability that...
-
Explain the difference between the one-tailed and two-tailed versions of the Wilcoxon rank sum test for independent random samples.
-
Determine a hypothesis that the following data may address and perform a x2 test on the survey data. a. American Idol should be broadcast before 9 p.m. Agree 40 Neutral 35 Disagree 25 100 b....
-
Chaz Corporation has taxable income in 2023 of $1,312,000 for purposes of computing the 179 expense and acquired the following assets during the year: Asset Placed in Service Basis Office furniture...
-
Drysdale Co. (a U.S. firm) is considering a new project that would result in cash flows of 5 million Argentine pesos in one year under the most likely economic and political conditions. The spot rate...
-
Explain which functional group(s) is present in the compound that has this IRspectrum: 100 - 50 1706 S00 1000 1500 2000 2500 3000 4000 3500 Wavenumber (cm
-
Suggest a possible structure for a compound with the formula C5H12O that has the following IR spectrum and explain yourreasoning: 80 60 40 20 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm)
-
What are the three levels at which statutes governing health information are found?
-
Your friend Amber has approached you seeking advice concerning two investment opportunities that she is presently considering. Her classmate Simone has asked her for a loan of $5,000 to help...
-
Please read the following carefully. For each question on the exam, you should assume that: 1. unless expressly stated to the contrary, all events occurred in ?the current taxable year;? 2. all...
-
The pulse rates of 152 randomly selected adult males vary from a low of 37 bpm to a high of 117 bpm. Find the minimum sample size required to estimate the mean pulse rate of adult males. Assume that...
-
Can I get clear explanation how to work these. Thanking you in advance. 1. A rod 12.0 cm long is uniformly charged and has a total charge of -23.0 uC. Determine the magnitude and direction of the...
-
Poll Results in the Media USA Today provided results from a survey of 1144 Americans who were asked if they approve of Brett Kavanaugh as the choice for Supreme Court justice. 51% of the respondents...
-
As a Chairperson leading a club what measures would you take to ensure your club gained Clubmark? How would you manage any resistance from other committee members to the work needed for a successful...
-
Why is homeostasis defined as the "relative constancy of the internal environments? Does negative feedback or positive feedback tend to promote homeostasis?
-
JSTOR (Journal Storage) is a nonprofit organization that sells subscriptions for Internet access to back issues of scholarly journals in fields such as chemistry, literature, business, and economics....
-
Identify the amino acid that is a catabolic precursor of each of the following ?-keto acids: (b) (a)
-
Identify the following intermediate in the citric acid cycle, and tell whether it has R or Sstereochemistry:
-
The following compound is an intermediate in the biosynthesis of one of the twenty common a-amino acids. Which one is it likely to be, and what kind of chemical change must take place to complete...
-
Cash from Operating Activities: ______________ Cash from Investing Activities: ______________ Cash from Financing Activities: ______________ Problem 2: Financial Ratios The GAP Macys 1 Current Ratio...
-
On January 1, 2021, Winky Enterprises issued 12% bonds dated January 1, 2021, with a face amount of $2,800,000. The bonds mature in 2030 (10 years). For bonds of similar risk and maturity, the market...
-
Using the following accounts and balances, prepare the stockholders' equity vection of the balance sheet. Pilty thousand shares of common stock are authorised, and 1,000 shares have been recoured,...

Study smarter with the SolutionInn App


