Question: For each of the ternary systems shown in figure, indicate whether: (a) Simple, countercurrent extraction, or (b) Countercurrent extraction with extract reflux, or (c) Countercurrent
For each of the ternary systems shown in figure, indicate whether:
(a) Simple, countercurrent extraction, or
(b) Countercurrent extraction with extract reflux, or
(c) Countercurrent extraction with raffinate reflux, or
(d) Countercurrent extraction with both extract and raffinate reflux would be expected to yield the most economicalprocess.
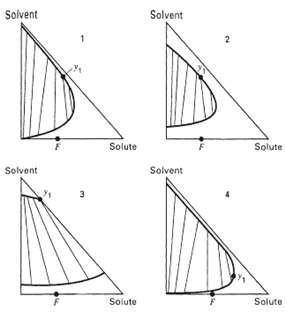
Solvent Solvent Solute Solute Solvent Solvent Solute Solute
Step by Step Solution
3.38 Rating (173 Votes )
There are 3 Steps involved in it
Subject Selection of extraction method Given Four ternary systems in Figure Diagra... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
37-E-C-E-S-P (314).docx
120 KBs Word File


