From Figure, find (a) The temperature at which water boils on a mountain where the atmospheric pressure
Question:
From Figure, find
(a) The temperature at which water boils on a mountain where the atmospheric pressure is 70kPa,
(b) The temperature at which water will boil in a container in which the pressure has been reduced to 0.5 atm, and
(c) The pressure at which water will boil at115oC.
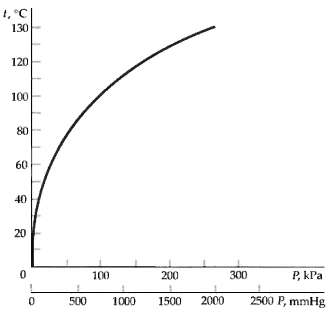
Transcribed Image Text:
1, °C 130 120 100 80 60 40 20 P, kPa 100 200 300 2500 P, mmHg 2000 500 1000 1500
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a At 70 kPa the boilin...View the full answer

Answered By

Danish Sohail
My objective is to become most reliable expert for clients. For last 10 years I have been associated with the field of accounting and finance. My aim is to strive for best results and pay particular attention to client needs. I am always enthusiastic to help clients for issues and concerns related to business studies. I can work on analysis of the financial statements, calculate different ratios and analysis of ratios. I can critically evaluate stock prices based on the financial analysis and valuation for companies using financial statements of the business entity being valued with use of excel tools. I have expertise to provide effective and reliable help for projects in corporate finance, equity investments, financial accounting, cost accounting, financial planning, business plans, marketing plans, performance measurement, budgeting, economic research, risk assessment, risk management, derivatives, fixed income investments, taxation, auditing, and financial performance analysis.
4.80+
78+ Reviews
112+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Thermodynamics questions
-
The temperature at which water boils (the boiling point) depends on elevation: The higher the elevation, the lower the boiling point will be. At sea level, water boils at 212F; at an elevation of...
-
The temperature at which water starts to boil is also linearly related to barometric pressure. Water boils at 212F at a pressure of 29.9 in Hg (inches of mercury) and at 191F at a pressure of 28.4 in...
-
A manufacturer of candy must monitor the temperature at which the candies are baked. Too much variation will cause inconsistency in the taste of the candy. Past records show that the standard...
-
Consider the pistoncylinder arrangement shown in the sketch below. The gas forces on each side of the piston, assuming there are no frictional forces at the pistoncylinder interface, balance the...
-
Steve Drake sells a rental house on January 1, 2016, and receives $120,000 cash and a note for $45,000 at 10 percent interest. The purchaser also assumes the mortgage on the property of $35,000....
-
Describe the circumstances in which the three major styles of leadership might be used effectively. Which would you use and why?
-
Latitude Energy decided to explore some acreage in Texas before acquiring any leases. Latitude acquired shooting rights only on 15,000 acres owned by Mr. T for \($0.10/acre. Latitude\) obtained...
-
Chris Zulliger was a chef at the Plaza Restaurant in the Snowbird Ski Resort in Utah. The restaurant is located at the base of a mountain. As a chef for the Plaza, Zulliger was instructed by his...
-
Question 6 Use the 2015 tax table to find the income tax for a taxpayer with taxable income of $91,124 filing as head of household. Use the 2015 tax table to find the income tax for a taxpayer with...
-
Yonica Petroleum is a global manufacturer of specialty chemicals that are made from the waste products of the petroleum industry. Yonica in effect recycles a good portion of the waste from the...
-
(a) Calculate the volume of 1 mol of steam at 100 o C and a pressure of 1 atm, assuming that it is an ideal gas. (b) Find the temperature at which the steam will occupy the volume found in part (a)...
-
The van der Waals constants for helium are a = 0.03412 L2atm/mol2 and b = 0.0237 L/mol. Use these data to find the volume in cubic centimeters occupied by one helium atom and to estimate the radius...
-
Okay Optical, Inc. (OOI), began operations in January, selling inexpensive sunglasses to large retailers like Walgreens and other smaller stores. Assume the following transactions occurred during its...
-
In a pie chart, if a category has a relative frequency of 30%, then its sector takes up 30% of the circle. In Exercises 912, determine whether the statement is true or false. If the statement is...
-
The_______________ of a class is the sum of the frequencies of that class and all previous classes. In Exercises 58, fill in each blank with the appropriate word or phrase.
-
What is the Duhamel integral? What is its use?
-
Fill in the Blank. The response of a single-degree-of-freedom system to a unit ____________ impulse response function.
-
A machine was acquired three years ago at \($850,000.\) Its life was estimated at five years, and its residual value after five years at \($50,000\) . a. What is its current book value? b. If the...
-
What do you believe is the most important issue facing sport management today?
-
How has the too-big-to-fail policy been limited in the FDICIA legislation? How might limiting the too-big-to-fail policy help reduce the risk of a future banking crisis?
-
When a pure substance melts, it (a) Undergoes a chemical change. (b) Changes state. (c) Changes from a liquid to a solid. (d) Sublimes.
-
The following measurements were made on a Thyrite resistor: (a) Graph V ab as a function of I. (b) Does Thyrite obey Ohm's law? How can you tell? (c) Graph the resistance R = Vab/I as a function of...
-
The following measurements of current and potential difference were made on a resistor constructed of Nichrome wire: (a) Graph Vab as a function of I. (b) Does Nichrome obey Ohm's law? How can you...
-
A resistor with a 15.0-V potential difference across its ends develops thermal energy at a rate of 327 W. (a) What is its resistance? (b) What is the current in the resistor?
-
An adjusting entry that credits income taxes payable is an example of: Group of answer choices deferred expenses deferred revenue accrued expenses accrued revenue
-
Page(s) 359-361 11.1. Explain the purpose of the state budget and what is typically included The budget of the state of Texas consists of five parts, representing different revenue sources and...
-
Crane Company invests $6.01 million in a mine that is estimated to have 8 million tonnes of ore and a $250000 residual value. In the first year, 23400 tonnes are extracted but only 7700 tonnes are...

Study smarter with the SolutionInn App


