If methanol rather than water is added at the end of a Hell-Volhard-Zelinskii reaction, an ester rather
Question:
If methanol rather than water is added at the end of a Hell-Volhard-Zelinskii reaction, an ester rather than an acid is produced. Show how you could carry out the following transformation, and propose a mechanism for the ester-formingstep.
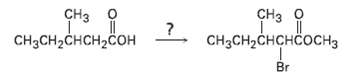
Transcribed Image Text:
CHз о CHз о сHзCH-CHCHCOСH3 CH3CH-CHCH-COн Br
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (17 reviews)
OH H CHCH3CHCH3 0 PBr3 Br formation of acid bromide HCH3 H Br Br H CHCH3CHCH3 enoliz...View the full answer

Answered By

Rukhsar Ansari
I am professional Chartered accountant and hold Master degree in commerce. Number crunching is my favorite thing. I have teaching experience of various subjects both online and offline. I am online tutor on various online platform.
5.00+
4+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How much mass should be at the end of a spring (k = 100 N/m) in order to have a period of 2.0 s?
-
Show how you could prepare each of the following compounds. Use the starting material indicated along with ethyl acetoacetate or diethyl malonate and any necessary inorganic reagents. Assume also...
-
The gate at the end of a 1-m-wide freshwater channel is fabricated from three 125-kg, rectangular steel plates. The gate is hinged at A and rests against a frictionless support at D. Knowing that...
-
Determine the maximum weight of the crate so that the tension developed in any cable does not exceed 450lb. 2 ft 1ft 2 f 2 ft,
-
Why are reports indispensable documents in business?
-
Consider a base band antipodal PAM system with channel bandwidth of 5 kHz and a desired data rate of 20 kbps. (a) What is the required value for M? (b) What value of E d / N 0 in dB will give a bit...
-
29. How does the tax treatment of a partial liquidation differ from a stock redemption?
-
A partial amortization schedule for a ten-year note payable that Muro Co. issued on January 1, 2012, is shown here: Required a. What rate of interest is Muro Co. paying on the note? b. Using a...
-
Common-Size Financial Statement Analysis Using Excel Completing this activity will help you learn to: 1. create Excel formulas using cell references. 2. create absolute cell references to perform...
-
One of Utility Company plans to install temporary power supply cables from existing Electrical Sub-station No. 210 to a temporary firewater pump located in existing seawater intake area. In view of...
-
Show how you might prepare 1-penten-3-one from 3-pentanone.
-
Identify the most acidic hydrogens in each of the following molecules: (a) CH3CH2CHO (b) (CH3) CH3CCOCH3 (c) CH3CO2H (d) Benz amide (e) CH3CH2CH2CN (f) CH3CON (CH3)2
-
Real mechanical systems may involve the deflection of nonlinear springs. In Fig. P6.20, a block of mass m is released a distance h above a nonlinear spring. The resistance force F of the spring is...
-
Select a qualitative research design to establish whether the strike by the federal workers in Canada may have been caused by a leadership
-
There is a single loop circuit with three resistors as follows. Take R-D 2, R= D/2 2 and R= D/3 2 and connect to 12 V battery. a. What can you say about the connection of R., R2 and Rs, Explain the...
-
1:This week we learned about Team Communication and Difficult conversation. Reflect on your own experience of a difficult conversation that you had to handle. Explain what happened and how you...
-
What is the value chain of Doordash? Name their primary and support activities.
-
Blue Water Clinic is a mobile field medical facility that has been offering Covid-19 vaccination services to rural communities in southwest Ontario under the supervision of medical authorities. With...
-
Construct corporate scenarios to evaluate strategic options AppendixLO1
-
What are the typical record-at-a-time operations for accessing a file? Which of these depend on the current file record?
-
How is the number of hybrid orbitals related to the number of standard atomic orbitals that are hybridized?
-
Explain how to use NMR spectroscopy to differentiate the isomers within each of the following sets. 1-bromo-4-ethylbenzene (Fig. 16.3) and (2-bromoethyl) benzene (BCH2CH2Ph) Fig. 16.3 benzylic...
-
Explain how to use NMR spectroscopy to differentiate the isomers within each of the following sets. 1-bromo-4-ethylbenzene (Fig. 16.3) and (2-bromoethyl) benzene (BCH2CH2Ph) Fig. 16.3 benzylic...
-
Would l-methoxynaphthalene nitrate more rapidly or more slowly than naphthalene at carbon-5 Explain your reasoning.
-
*please calculate irr in excel
-
Which of the following would not be a period cost? Research and development Direct materials Office supplies Advertising costs
-
\ table [ [ Activity Cost Pool,Activity Measure,Total Cost,Total Activity ] , [ Machining , Machine - hours,$ 3 3 0 , 0 0 0 , 1 5 , 0 0 0 MHs ] , [ Machine setups,Number of setups,$ 3 0 0 , 0 0 0 , 5...

Study smarter with the SolutionInn App


