In Figure, suppose Br2 adds to the alkene from the bottom, rather than from the top as
Question:
In Figure, suppose Br2 adds to the alkene from the bottom, rather than from the top as shown. Analyze the stereochemistry of the reaction in this case and explain which products areformed.
Transcribed Image Text:
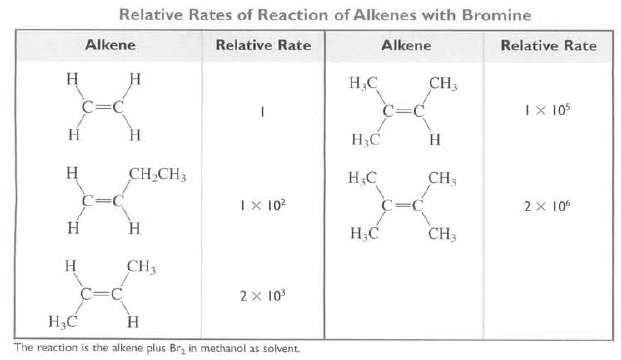
Relative Rates of Reaction of Alkenes with Bromine Alkene Relative Rate Alkene Relative Rate Н CH3 Н.С C=C IX 105 Н Н Н С Н Н CH CH3 CH3 НС C=C IX 10? 2 x 10 Н Н Н.С CH3 CH3 Н 2 x 10 H3C н The reaction is the alkene plus Br, in mathanol as solvent.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
If Br 2 adds from the bottom rather than the as shown in Figur...View the full answer

Answered By

Nandana Wijayarathna
I am a highly experienced writer in several areas,
Business management
Information technology
Business administration
Literature
Biology
Environmental science
History
4.50+
161+ Reviews
399+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The rate law for addition of Br2 to an alkene is first order in Br2 and first order in the alkene. Does this fact prove that the mechanism of addition of Br2 to an alkene proceeds in the same manner...
-
The gas-phase reaction between Br2 and H2 to form HBr is assumed to proceed by the following mechanism: a. Under what conditions does the rate law have the form rate = k[Br2]? b. Under what...
-
From what you know about the stereochemistry of alkene addition reactions, predict the configurations of the products that would be obtained from the reaction of 2-butyne with the following: a. One...
-
0 3 S 25 cos(x) sin (x) dx -T3
-
Graywall, CPA, serves as a trustee of the CureCancerNow Foundation, a charitable foundation that is dedicated to cancer research activities. For years, he has been the loyal friend and advisor to...
-
Cantu Company is a rapidly growing start-up business. Its recordkeeper, who was hired nine months ago, left town after the companys manager discovered that a large sum of money had disappeared over...
-
Understanding inventory cost data Data relevant to Job No. QV1173 are: An analysis of the production data indicate a number of additional items as follows: a. In addition to the 200 good units, there...
-
Pension Plans The Company maintains defined benefit, non- contributory retirement plans for substantially all of its employees not participating in multi- employer pension plans. Safeway recognizes...
-
. ) . ) . . . . (GAAP) IFRS ( ). . . )
-
Atwood Company has an opportunity to produce and sell a revolutionary new smoke detector for homes. To determine whether this would be a profitable venture, the company has gathered the following...
-
Show the structures of A, B, C, and D in the following reactionsscheme: D Optically inactive H,SO. H,O B Pt C,H14 C,H12 Optically Optically inactive active 1) Hg(O,CCH3)2, H20 2) NaBH4, NAOH...
-
Explain which of the three products shown in is formed when 1-butene reacts with HCI.
-
What is the probability of winning a 5/30 lottery game where 5 numbers are picked from a range of 30?
-
State the principle of superposition.
-
State the conditions of static equilibrium.
-
Can an audit be conducted efficiently without specifying materiality before starting specific audit procedures? Explain. What is the risk to the audit if materiality is not specified in advance of...
-
What is equivalent offset inertia force?
-
Define the accounting concept of materiality. How does the concept of the reasonable person affect the auditors determination of materiality?
-
Define infinite recursion and discuss ways to avoid it.
-
Describe basic managerial approaches to implementing controls and how these are implemented.
-
A first-order reaction has rate constants of 4.6 10 -2 s -1 and 8.1 10 -2 s -1 at 0C and 20.C, respectively. What is the value of the activation energy?
-
To what classes do the following enzymes belong? (a) Pyruvate decarboxylase (b) Chymotrypsin (c) Alcohol dehydrogenase
-
Identify the following aminoacids: (a) (b) (c)
-
Give the sequence of the following tetra peptide (yellow =S):
-
Analyze the standard costing system employed by Tesla, Inc. For a specific product, the standard cost per unit is $50, comprising $30 for direct materials and $20 for direct labor. During the last...
-
Transfer Pricing at Microsoft Microsoft is evaluating its transfer pricing strategy. Values: Item Amount Production Cost $80 per unit Profit Margin 30% Requirements: Determine the minimum transfer...
-
Luna was recruited to serve as the Chief Financial Officer (CFO) of Alpha Corporation, a publicly-traded company in the healthcare industry, effective March 1, 2022. For the fiscal year 2023, Alpha...

Study smarter with the SolutionInn App


