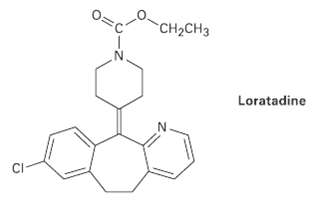
Loratadine, marketed as an antiallergy medication under the name Claritin, has four rings, eight double bonds, and
Question:
Loratadine, marketed as an antiallergy medication under the name Claritin, has four rings, eight double bonds, and the formula C22H? C1N2O2. How many types of hydrogen does loratadine have? (Calculate your answer; don?t count hydrogens in the structure.)
Transcribed Image Text:
CH2CH3 Loratadine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
Solve this problem in the same way as we solved problems A C22 hydr...View the full answer

Answered By

Sarah Khan
My core expertise are:
-_ Finance
-_ Business
-_ Management
-_ Marketing Management
-_ Financial Management
-_ Corporate Finance
-_ HRM etc...
I have 7+ years of experience as an online tutor. I have hands-on experience in handling:
-_ Academic Papers
-_ Research Paper
-_ Dissertation Paper
-_ Case study analysis
-_ Research Proposals
-_ Business Plan
-_ Complexed financial calculations in excel
-_ Home Work Assistance
-_ PPT
-_ Thesis Paper
-_ Capstone Papers
-_ Essay Writing etc...
5.00+
91+ Reviews
92+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many types of atoms can you expect to find in a pure sample of any element?
-
Diazepam, marketed as an antianxiety medication under the name Valium, has three rings, eight double bonds, and the formula C 16 H ?? C1N 2 O. How many types of hydrogen does diazepam have?...
-
How many types of hydrogen does each of the following compounds have? (a) C8H? O2, has two rings and one double bond (b) C7H? N, has two double bonds (c) C911? NO, have one ring and three double bonds
-
Select appropriate letter to each of its related definition in the list given below. Definition A. Economic resources to be used or turned into cash within one year. B. Reports assets, liabilities,...
-
Several people have commented that this chapter deals with the "nuts and bolts" of leadership. What makes them say that?
-
What activities qualify for the qualified production activities deduction?
-
Material price, mix, and yield variances Lemon-Yellow makes a powdered lemonade mix, which it sells in two pound cans. The standard material costs are presented below: Quantity Price Total Material...
-
Douglas Corporation had 120,000 shares of stock outstanding on January 1, 2010. On May 1, 2010, Douglas issued 60,000 shares. On July 1, Douglas purchased 10,000 treasury shares, which were reissued...
-
( i ) Required: Compute the total prime costs for both Garcon Company and Pepper Company. Compute the total conversion costs for both Garcon Company and Pepper Company. Complete this question by...
-
A junior CPA at Depaul, CPAs currently is working as an audit team member on the audit of General Molecular. This CPA is about to search for a different position of employment If this individual is...
-
Calculate the degree of unsaturation in the following formulas, and draw five possible structures for each: (a) C10H16 (b) C8H8O (c) C7H10C12 (d) C10H16O2 (e) C5H9NO2 (f) C8H10C1NO
-
Name the following alkenes: ) CH2H2CH {c) CH-CH (a) H (b) -CH CHCH2CH3 CH H%3H2H H3C (9) H%3D%3H3 (e) CH (d) C=C 23DCHCHCH CH CH CH CHCH2CH2
-
1. Select at least two other firms that you believe have crossed the chasm. Briefly summarize how the firms rolled out their products (or services) and what you believe their key steps were in...
-
George is interested in buying one of the two local businesses on sale: a Coffee Shop or a Shoe Store. The cashflows of both the businesses are shown below. George can borrow from a bank at a rate of...
-
Bank CO approved a loan application of Client ME on January 1, 20A for P3,500,000. Client ME is required to pay 15% interest annually every December 31, from 20A until the principal is extinguished....
-
Brad's Fish and Chips Restaurant is booming and as such he needs an automated beer pouring machine. He is looking at two used models, one with a life span of 4 years which costs $28,000 and another...
-
discuss the molecular regulation of sex determination and differentiation, including the roles of sex chromosomes, sex-determining genes, and hormone signaling pathways in specifying gonadal fate and...
-
2 Company produces product cheese and butter jointly. During October production data as follows: 3 Cheese Butter 4 Gallons at the splitt off point 100,000 150,000 5 October production of final...
-
Develop an integer programming model for the resource allocation problem in Figure 7.47, assuming that daily resource availability is 5 units. Use LINDO or any other integer programming software to...
-
How much more interest will be earned if $5000 is invested for 6 years at 7% compounded continuously, instead of at 7% compounded quarterly?
-
Spilling room-temperature water over your skin on a hot day cools you down. Spilling room-temperature vegetable oil over your skin on a hot day does not. Explain the difference.
-
Why isn't the alkene starting material in Eq. 4.43 part of the equilibrium mixture?
-
Isopropyl alcohol is produced commercially by the hydration of propene. Show the mechanistic steps of this process. If you do not know the structure of isopropyl alcohol, try to deduce it by analogy...
-
Give the structures and the IUPAC substitutive names of the isomeric alkenes with the molecular formula CuH,, containing four carbons in their principal chain.
-
Journalizing And Posting Payroll Entries Cascade Company has four employees. All are paid on a monthly basis. The fiscal year of the business is June 1 to May 3 1 . The accounts kept by Cascade...
-
Carlton holds undeveloped land for investment. His adjusted basis in the land is $ 2 0 0 , 0 0 0 , and the FMV is $ 3 2 5 , 0 0 0 . On November 1 , 2 0 2 1 , he exchanges this land for land owned by...
-
Calculate Current Ratio: Requirements: Given the balance sheet data: Current Assets: $200,000 Current Liabilities: $80,000 Compute the current ratio. Provide the calculated current ratio.

Study smarter with the SolutionInn App


