Phenol, C6H5OH, is a stronger acid then methanol, CH3OH, even though both contains an O ? H
Question:
Phenol, C6H5OH, is a stronger acid then methanol, CH3OH, even though both contains an O ? H bond. Draw the structures of the anions resulting from loss of H+ from phenol and methanol, and use resonance structures to explain the difference in acidity.
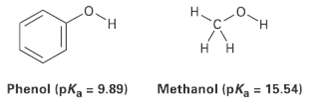
Transcribed Image Text:
H. H. Methanol (pKa = 15.54) = 9.89) Phenol (pK
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 73% (15 reviews)
H 66666 HH When p...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Related Video
In this video, A mixture of methanol and air in a large polycarbonate bottle is ignited. The resulting rapid combustion reaction, often accompanied by a dramatic ‘whoosh’ sound and flames, demonstrates the large amount of chemical energy released in the combustion of alcohol
Students also viewed these Organic Chemistry questions
-
Explain the following observations: (a) HNO3 is a stronger acid than HNO2; (b) H2S is a stronger acid than H2O; (c) H2SO4 is a stronger acid than HSO4-; (d) H2SO4 is a stronger acid than H2SeO4; (e)...
-
Explain the following observations: (a) HCl is a stronger acid than H2S; (b) H3PO4 is a stronger acid than H3AsO4; (c) HBrO3 is a stronger acid than HBrO2; (d) H2C2O4 is a stronger acid than HC2O4-;...
-
Use resonance formulas to explain why polyacetylene has delocalized molecular orbitals extending over the length of the molecule, whereas the following molecule does not. HHHHH
-
Why should one-time write-offs of fixed capital be used in absorption cost systems?
-
Samuel and Annamaria are married, file a joint return, and have three qualifying children. In 2016, they earn wages of $34,000 and no other income. Determine the amount of their earned income credit...
-
A cylindrical barrel s feet in diameter of weight w lb is floating in water as shown in the following figure (a). After an initial depression the barrel exhibits an up and down bobbing motion along a...
-
Assume that you must make future value estimates using the future value of 1 table (Table B.2). Which interest rate column do you use when working with the following rates? 1 8% compounded quarterly...
-
Weinberg Canning produces fillet, smoked salmon, and salmon remnants in a single process. The same amount of disposal cost is incurred whether a product is sold at split-off or after further...
-
Schefter Mining operates a copper mine in Wyoming Acquisition, exploration, and development costs totaled $8.6 million Extraction activities began on July 1 2018. After the copper is extracted in...
-
You decided to run an experiment - improve current CTA on the in-app pricing page (1 experimental variation and one control group). Each month the pricing page is seen by 16,000 users. 800 of those...
-
We?ll see that organic molecules can be classified according to the functional groups they contain, where a functional group is a collection of atoms with a characteristics chemical reactivity. Use...
-
Monobromination of toluene gives a mixture of three bromotoluene products. Draw and name them.
-
Suppose that in 10 years the states have the following population and 30 seats are apportioned. If the seats are reapportioned, does the population paradox occur using Hamiltons method? A country has...
-
Implement the nearest neighbor algorithm in the programming language of your choice. The algorithm should work with vectors of up to 10 integer values and allow up to 10 integer classifications. By...
-
Use the operators described in Section 16.2.4 and the STRIPS method to solve the block world planning problem shown in Figure 16.11. The first state shown is the start state and the second state is...
-
Implement a Bayesian belief network in the programming language of your choice to represent a subject in which you are interested (for example, you might use it to diagnose medical conditions from...
-
Researchers have measured the acceleration of racing greyhounds as a function of their speed; a simplified version of their results is shown in Figure P4.67. The acceleration at low speeds is...
-
If the rate at which energy is dissipated by resistor 1 in Figure P31.86 is \(2.5 \mathrm{~W}\), and \(R_{1}=10 \Omega, \mathscr{E}_{1}=12 \mathrm{~V}\), and \(\mathscr{E}_{2}=6 \mathrm{~V},\) (a)...
-
What is x? x . . . . . . . . . . . . . . forces management to set clear objectives and develop their staff. Senior auditors may spend hours on an obscure project that provides no end-product. x . . ....
-
Prove that if Σ an is absolutely convergent, then a. an
-
Using the Redlich-Kwong equation of state, compute the following quantities for nitrogen at 298.15 K. a. The difference C P C V as a function of pressure from low pressures to very high pressures b....
-
Arrange these compounds in order of increasing SN2 reaction rate: CI Br CI Br
-
Arrange these compounds in order of increasing SN1 reaction rate: Ph-Br Br + Br CI
-
Show the products of these reactions and explain whether each would follow an SN1 or an SN2 mechanism: a) C) B CI + OH Br + SH DMF CHOH HO Br + HO CHOH HO b) d) f) CI + HO CH,OH OTS + CH0 Br + CHCO...
-
Which of the following journal entries will record the payment of a $1,500 salaries payable originally incurred for Salaries Expense? Select one: A. Debit Salaries Expense; credit Salaries Payable B....
-
What is the definition of substantially appreciated inventory? A. Inventory with a FMV greater than its basis B. Inventory and unrealized receivables with a FMV greater than their basis C. Inventory...
-
Case Products manufactures two models of DVD storage cases: regular and deluxe. Presented is standard cost information for each model: Cost Components Regular Deluxe Direct materials Lumber 2 board...

Study smarter with the SolutionInn App


