Propose a mechanism to account for the following reaction: C CH2CI AICI3
Question:
Propose a mechanism to account for the following reaction:
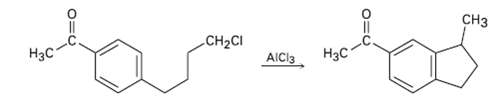
Transcribed Image Text:
Cнз CH2CI Нас Нзс AICI3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
H CHCl love formation of H3C0 H3C HC CIAICI3 CH3 HA loss of H AICI 3 CH3 ...View the full answer

Answered By

John Kago
Am a processional practicing accountant with 5 years experience in practice, I also happens to have hands on experience in economic analysis and statistical research for 3 years. am well conversant with Accounting packages, sage, pastel, quick books, hansa world, etc, I have real work experience with Strata, and SPSS
4.70+
31+ Reviews
77+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism to account for the reaction of benzene with 2, 2, 5, 5- tetra methyl tetra hydrofuran. H2SO4
-
Propose a mechanism to account for the formation of 3, 5-dimethylpyrazole from hydrazine and 2, 4-pcntancdionc. Look carefully to see what has happened to each carbonyl carbon in going from starting...
-
Propose a mechanism to account for the fact that heating 1-deuterioindene scrambles the isotope label to all three positions on the five-memberedring. 1-Deuterioindene
-
What is the result of the following? A. [Natural History, Science] B. [Natural History, Science, Art] C. The code does not compile. D. The code compiles but throws an exception at runtime. } import...
-
Morgan (age 45) is single and provides more than 50% of the support of Rosalyn (a family friend), Flo (a niece, age 18), and Jerold (a nephew, age 18). Both Rosalyn and Flo live with Morgan, but...
-
The data values below represent the closing prices of the 20 most actively traded stocks on the NASDAQ Stock Exchange (rounded to the nearest dollar) on May 2, 2014. a. Sketch a dot plot or construct...
-
Refer to the balance sheet of Circuit City in Appendix A. How can you tell that Circuit City uses the consolidated method of accounting? AppendixLO1
-
An investor bought common stock of Microsoft Corporation on three occasions at the following prices. Calculate the average price per share at which the investor bought these shares. Date January 2009...
-
Match the following elements of internal control: Prompts Answers Control Environment Risk Assessment Control Procedures Monitoring Information and Communication
-
Mary is a coworker in your agency. She has been a valuable employee to your group and one of the most respected experts in her field. You notice lately, though, that she is more reserved and is...
-
P-Bromotoluene reacts with potassium amide to give a mixture of m- and p-methyl aniline. Explain.
-
In the Gatterman-Koch reaction, a formyl group (?CHO) is introduced directly onto a benzene ring. For example, reaction of toluene with CO and HCl in the presence of mixed CuCl/AlCl 3 gives...
-
CVP missing data. Management of Beijing Manufacturing has performed cost studies and projected the following annual costs based on 200,000 units of production and sales: a. Compute Beijing's unit...
-
You are the manager of internal audit of Coverit Corporation, a large insurance company. One day you receive an urgent letter from the controller expressing his concerns about some organizational...
-
Daintree Ltd. is a large retailer that operates department stores in all major cities throughout Australia. Recently it has expanded its operations into Southeast Asia. Although each store operates...
-
Draw two points P and Q. Then sketch PQ. Add a point R on the ray so that Q is between P and R. C D A B FL E
-
Hypothesis testing and testing claims with confidence intervals are two different approaches that lead to the same conclusion. In the following activities, you will compare and contrast those two...
-
The following system of periodic tasks is scheduled and executed according to a cyclic schedule. Draw an execution trace (timeline) showing two occurances of each task. Ti ei Pi 1 8 T2 4 15 T3 3 20...
-
How might a Starbucks store manager use situational leadership theory? Path-goal theory? Transformational leadership?
-
Per Bag Direct materials: 25 pounds of CWhiz-2000 @ $0.08/lb. = $ 2.00 Direct labor: 0.05 hour @ $32.00/hr. = $ 1.60 The company manufactured 100,000 bags of Cheese-Be-Good in December and used...
-
Electrical power is to be produced from a steam turbine connected to a nuclear reactor. Steam is obtained from the reactor at 540 K and 36 bar, the turbine exit pressure is 1.0 bar, and the turbine...
-
Show the resonance structures for the conjugate base of phenol.
-
The pKa for phenol is 10, the pKa for ethanol is 16, and the pKa for carbonic acid (H2CO3) is 6.35. Complete these equations and predict whether the reactants or the products are favored...
-
Provide names for thesecompounds: a) CH;CH,CH,CH,CH,CH b) CH f) CH,CCH,CCH, d) CI g) h)
-
solve this plz Alba Company is considering the introduction of a new product. To determine the selle price of the product you have The direct material permit The direct labor per unit The variable...
-
Calculate the current ratio collection period for accounts receivable, inventory turnover, gross margin percentage, and return on equity for 2014 and 2015 for the Jordan Corporation. Do not average....
-
A company received $11,000 cash in exchange for 200 shares of the companys common stock. What would the effect of this transaction on the current years accounting equation? Select one: A. No effect...

Study smarter with the SolutionInn App


