Rank the following substituted phenols in order of increasing acidity, and explain your answer: CH
Question:
Rank the following substituted phenols in order of increasing acidity, and explain your answer:
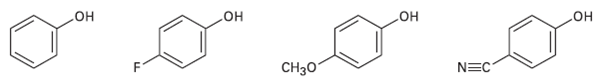
Transcribed Image Text:
CHзо но но но он
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
Remember that electronwithdrawing groups stabilize phenox...View the full answer

Answered By

BETHUEL RUTTO
Hi! I am a Journalism and Mass Communication graduate; I have written many academic essays, including argumentative essays, research papers, and literary analysis. I have also proofread and written reviews, summaries and analyses on already finished works. I am eager to continue writing!
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Rank the following complex ions in order of increasing wavelength of light absorbed. Co(H20+ CoCN)-, Col, Colen)
-
Rank the following in order of increasing acidity for the indicated hydrogen atoms (bold) (1 = least acidic; 4 = most acidic). (a) (b) (c) O C OCH3 . CH3 H3C
-
Rank the following five compounds in order of increasing acid strength: 2-chloroethanol, p-chlorophenol, p-methylphenol, ethanol, and phenol.
-
Which of the following is not an element of the fraud triangle? (a) Rationalization. (b) Financial pressure. (c) Segregation of duties. (d) Opportunity.
-
How does environmental analysis at the domestic level differ from global analysis?
-
At the beginning of 2011, Huffer Corporation had total assets of $226,800, total liabilities of $84,200, common stock of $80,000, and retained earnings of $62,600. During 2011, Huffer had net income...
-
Use supply and demand curves to illustrate how a central bank conducts monetary policy using open market operations, a discount window and altering reserve levels.
-
1. Draw the bricks and mortar process stages by which hard copy books are created, distributed, and sold in retail stores. How does each player in the value chain make money? (You can use the...
-
Brian Walker manufactures and sells homemade wine, and he wants to develop a standard cost per litre. The following are required for production of a 350-litre batch: 84.00 litres of grape concentrate...
-
Prairie Grains Cooperative wants to invest $45 000 in a short-term deposit. The bank offers 1.3% interest for a one-year term and 1.1% for a six-month term. (a) How much would Prairie Grains receive...
-
Benzoquinone is an excellent dienophile in the DielsAlder reaction. What product would you expect from reaction of Benzoquinone with 1 equivalent of 1, 3-butadiene from reaction with 2 equivalents of...
-
Benzyl chloride can be converted into benzaldehyde by treatment with nitro methane and base. The reaction involves initial conversion of nitro methane into its anion, followed by SN2 reaction of the...
-
Suppose that auto workers in South Korea can pro-duce components used to make cars at the rate of six sets of components per worker per day and can assemble cars at the rate of four cars per worker...
-
_____ occurs when the level of difficulty for an organization to provide accommodations, determined by looking at the nature and cost of the accommodation and the overall financial resources of the...
-
Briefly explain the difference between repair maintenance, adaptive maintenance, and perfective maintenance. Why are auditors concerned with controls over all types of program maintenance?
-
Briefly describe the relationship between desk checking, structured walkthroughs, and design and code inspections.
-
Briefly explain the difference between static analysis tests and dynamic analysis tests. For each type of test, give two types of automated tools that programmers might use to help them conduct...
-
Briefly explain the role of the librarian in a chief programmer team. What duties does the librarian have with respect to the program production library? How does the librarian role inhibit...
-
The work environment includes these factors: a) Lighting, noise, temperature, and air quality b) Illumination, carpeting, and high ceilings c) Enough space for meetings and videoconferencing d)...
-
TRUE OR FALSE: 1. Banks with a significantly large share of fixed-interest rate home loans are less exposed to interest rate risks. 2. Although Australian banks are pretty big, they are not...
-
Show that the formation of NaCl 2 (s) is very unfavorable; that is, fH[NaCl 2 (s)] is a large positive quantity. Assume that the lattice energy for NaCl 2 would be about the same as that of MgCl 2 ,...
-
Classify the following solvents as being protic or aprotic: formic acid, HCO2H; acetone, CH3COCH3; acetonitrile, CH3CN; formamide, HCONH2; sulfur dioxide, SO2; ammonia, NH3; trimethylamine, N(CH3)3;...
-
Would you expect the reaction of propyl bromide with sodium cyanide (NaCN), that is, CH3CH2CH2Br + NaCN CH3CH2CH2CN + NaBr to occur faster in DMF or in ethanol? Explain your answer.
-
When tert-butyl bromide undergoes solvolysis in a mixture of methanol and water, the rate of solvolysis (measured by the rate at which bromide ions form in the mixture) increases when the percentage...
-
You are considering a stock investment in one of two firms (LotsofDebt, Incorporated and LotsofEquity, Incorporated), both of which operate in the same industry. LotsofDebt, Incorporated finances its...
-
Blooper Industries must replace its magnoosium purification system. Quick & Dirty Systems sells a relatively cheap purification system for $16 million. The system will last 4 years. Do-It-Right sells...
-
3 2 points Problem 7-1 (LO7.1) Based on the following data, calculate the items requested: Rental Costs Annual rent Buying Costs eBook Hint Print References $ 8,080 Annual mortgage payments Insurance...

Study smarter with the SolutionInn App


