Show the structures of alkenes that give the following products on oxidative cleavage with KMnO4 in acidic
Question:
Show the structures of alkenes that give the following products on oxidative cleavage with KMnO4 in acidic solution:
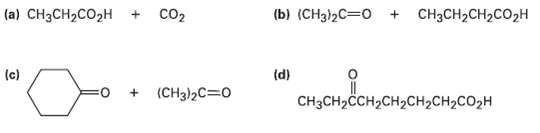
Transcribed Image Text:
(b) (CHз])2С—о + |(a) CHзCH2CO2н co2 CH3CH2CH2CO2H (d) (c) (CH3)2C=0 CнзCH-CCH2CH2CHCH2CO2H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
Remember that alkenes can give ketones carboxylic a...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In each of the following pairs, determine whether the two represent resonance forms of a single species or depict different substances. If two structures are not resonance forms, explain why. All the...
-
Suggest structures for alkenes that give the following reaction products. There may be more than one answer for some cases. H (b) CH CH3 /Pd CH2H2H2CH H2/Pd (d) CH Br CHH2CHCH (c) Br2 HCI ,...
-
Show the structures of all possible acid-catalyzed dehydration products of the following. If more than one alkene is possible, predict which one will be formed in the largest amount. a....
-
Consider the frequency demodulation scheme in Figure in which the incoming FM signal s (t) is passed through a delay line that produces a phase-shift of ?/2 radians at the carrier frequency ? c . The...
-
Given P(A) = .40, P(B) = .50, and P(A B) = .05. (a) Find P(A | B). (b) In this problem, are A and B independent?
-
Show that the least squares solution of Ax = b is not unique and solve the normal equations to find all the least squares solutions. 1 -3 ,b = 2 |A = 0 -1 -1 4
-
10. How is the tax treatment of restricted stock different from that of NQOs? How is it similar?
-
Presented below is financial information related to the 2017 operations of Sea Legs Cruise Company. Maintenance and repairs expense ....... $ 95,000 Utilities expense ............... 13,000 Salaries...
-
Within your study scoop of the Management Risk Financial course, explain : Ameen alswaeer
-
Define risk aversion in words without reference to probability distributions. If people are risk averse, why are lotteries so popular? Why are trips to Las Vegas popular?
-
Draw the structure of an alkene that yields only acetone, (CH3)2C = O, on ozonolysis followed by treatment with Zn.
-
Compound A has the formula C10H16. On catalytic hydrogenation over palladium, it reacts with only I molar equivalent of H2. Compound A also undergoes reaction with ozone, followed by zinc treatment,...
-
Outline the multiple, conflicting objectives of the annual report.
-
Woodland Wearables produces two models of smartwatches, the Basic and the Flash. The watches have the following characteristics:Basic Flash Selling price per watch$ is 270$ 460 Variable cost per...
-
Based on the information provided and recognizing the value of coordinating across its portfolio of businesses, how should LendingTree manage these newer businesses? * as more integrated units * as...
-
Trust Fund Worksheet Background An inter vivos trust was created by Isaac Posney. Isaac owned a large department store in Juggins, Utah. Adjacent to the store, Isaac also owned a tract of land that...
-
A popular theory is that presidential candidates have an advantage if they are taller than their main opponents. Listed are heights (in centimeters) of randomly selected presidents along with the...
-
Gracia Enterprises operates across five industries. Task 1 : After reviewing the information provided, determine which of the five operating segments are reportable based on the revenue test, asset...
-
Which costs are relevant for decisions to drop products LO.1
-
Find the image of x = k = const under w = 1/z. Use formulas similar to those in Example 1. y| y = 0 -21 -2 -1 -1, /1 12 T -1 -1 y= -2 x =0
-
What are the main properties of liquids (in contrast to gases and solids)?
-
Choose the alkyl halide(s) frorn the following list of C6H13Br isomers that meet each criterion below. (1) l-bromohexane (2) 3 -bromo-3 -methylpentane (3 ) I -bromo-2,2-dimethylbutane (4) 3 -bromo-2-...
-
Rank the following compounds in orcler of increasing SN2 reaction rate with KI in acetone. methyl bromide sec-butyl bromide 3-(bromomethyl)-3-methylpentane I-bromopentane -bromo-2-methylbutane
-
Rank the following compounds in orcler of increasing SN2 reaction rate with KI in acetone. methyl bromide sec-butyl bromide 3-(bromomethyl)-3-methylpentane I-bromopentane -bromo-2-methylbutane
-
XF Ltd. Is an expanding private company in the electric trade. Accounts preparing in January 2019 included the following information: Profit Statement for the year ended 31 st December 2018 Kshs.000...
-
Check On June 15, 2021, Sanderson Construction entered into a long-term construction contract to build a baseball stadium in Washington D.C., for $340 million. The expected completion date is April...
-
Q.1 Bassem Company purchased OMR420,000 in merchandise on account during the month of April, and merchandise costing OMR $350,000 was sold on account for OMR 425,000. Required: 1. Prepare journal...

Study smarter with the SolutionInn App


