You need to make a buffer whose pH is 7.0, and you can choose from the weak
Question:
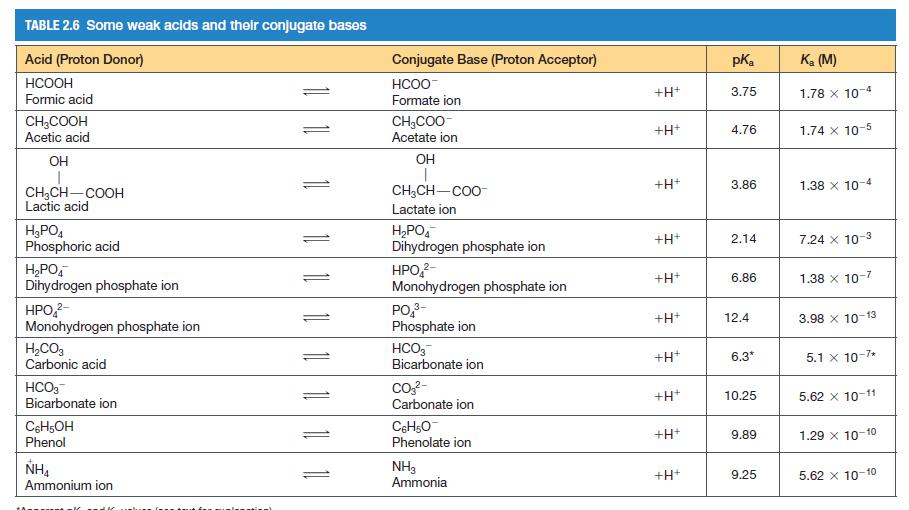
You need to make a buffer whose pH is 7.0, and you can choose from the weak acids shown in Table 2.6. Briefly explain your choice.
Transcribed Image Text:
TABLE 2.6 Some weak acids and their conjugate bases Acid (Proton Donor) HCOOH Formic acid CH3COOH Acetic acid OH CH₂CH-COOH Lactic acid H₂PO4 Phosphoric acid H₂PO4 Dihydrogen phosphate ion HPO ²- Monohydrogen phosphate ion H₂CO3 Carbonic acid HCO3 Bicarbonate ion C6H5OH Phenol NHA Ammonium ion 11 11 11 1 Conjugate Base (Proton Acceptor) HCOO Formate ion CH₂COO- Acetate ion OH T CH₂CH-COO Lactate ion H₂PO4 Dihydrogen phosphate ion HPO₂²- Monohydrogen phosphate ion PO ³- Phosphate ion HCO3 Bicarbonate ion CO3²- Carbonate ion C6H5O Phenolate ion NH3 Ammonia +H+ +H+ +H+ +H+ +H+ +H+ +H+ +H* +H+ +H+ pK₂ 3.75 4.76 3.86 2.14 6.86 12.4 6.3* 10.25 9.89 9.25 K₂ (M) 1.78 x 10-4 1.74 x 10-5 1.38 x 10-4 7.24 x 10-3 1.38 x 10-7 3.98 x 10-13 5.1 x 10-7* 5.62 X 10-11 1.29 X 10-10 5.62 x 10-10
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
To create a buffer solution with a pH of 70 you should choose a weak acid and its conjugate base tha...View the full answer

Answered By

Amit Choudhary
I'm new in this profession regarding online teaching but previously i used to teach students near my college. I am teaching on online platform since last year and got good support from the students. I'm teaching on platforms like chegg and vedantu and also at my home in free time.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Biochemistry Concepts And Connections
ISBN: 9780134641621
2nd Edition
Authors: Dean Appling, Spencer Anthony-Cahill, Christopher Mathews
Question Posted:
Students also viewed these Sciences questions
-
Analytics mindset Final Project 4 Gamification Part 1: Background You are the chief technology officer (CTO) of an international bank. A key component of your job is to manage risk within the bank...
-
Which of the following is NOT a process in feasibility study? a. Production b. Market Analysis c. Idea generation O d. Business Plan
-
Compare and contrast ROR using different definitions of airport assets, including revenue generator, public service, safety, and security infrastructure.
-
As an employer, what are steps you can take to avoid discrimination litigation?
-
How might the business plan change if the reader were an investor versus a potential management hire?
-
Describe how the genome of bacteriophage X174 is transcribed and translated.
-
Briefly describe the criticisms of the tort system that led to the enactment of automobile no-fault laws.
-
On December 31, 2012, Cathy Chen, a self-employed certified public accountant (CPA), completed her first full year in business. During the year, she billed $360,000 for her accounting services. She...
-
Target reported the following information for 2020: stockholders equity at January 1: 320,000 Net income: 20,000 dividends paid: 60,000 issuance of common stock to stockholders: 40,000 What is the...
-
Is RNA-binding enzyme RNase A more likely to have a pI of 9.2 or 5.0? Briefly explain your reasoning.
-
Cis-Vaccenate is an 18-carbon unsaturated fatty acid abundant in E. coli membrane lipids. Propose a metabolic route for synthesis of this fatty acid, in light of the fact that stearic acid, the C 18...
-
Evaluate the effectiveness of this proposal based on information you have studied in this chapter. Discuss the strengths and weaknesses of the proposal. Are there any items missing from the proposal...
-
$ 12,865 The Fiberglass Boat Company reported the following costs and expenses in May 2021: Factory utilities Direct labor $209,860 Depreciation on factory Sales staff salaries 134,520 equipment...
-
If the price of X is $26, the firm will (a) shut down in the short run. Wrong. The company will produce if Price>AVC at any level of Q. If Price>AVC the company makes some contribution to fixed...
-
b) Find Find b c) b 50m b 45%
-
Question 1: Differentiation and marketing strategy analysis (20 marks) a) b) c) d) With the new strategy unveiled to drive growth at Ford, please write a positioning statement for Ford. (3 marks) Is...
-
Briefly explain the concepts below a) How is a register built from flipflops? How is a counter built from flipflops? How many flipflops to use in each case? b) Describe how interrupts work in the...
-
A rod is negatively charged by rubbing it with fur. It is brought near another rod of unknown composition and charge. There is a repulsive force between the two. (a) Is the first rod an insulator or...
-
On August 31, 2012, the balances of the accounts appearing in the ledger of Wood Interiors Company, a furniture wholesaler, are as follows:Prepare the August 31, 2012, closing entries for Wood...
-
How is it advantageous for animals to convert ingested starch to glucose and then to incorporate the glucose into glycogen?
-
Suggest a reason or reasons why the Cori cycle takes place in the liver and in muscle.
-
Why would you expect to see that reactions of substrate cycles involve different enzymes for different directions?
-
compare asset sales to stock sales
-
Jane White has recorded the following sales figures for the last year of her business: January $35,645, February $35,456, March $31,270, April $32,129, May $34,456, June $35,356, July $36,218, August...
-
This budget represents the anticipated receipts and payment of cash during the budget period. The cash budget also called as Functional Budget. Cash budget is the most important of the entire...

Study smarter with the SolutionInn App


