(a) Use a spreadsheet program or graphing software and data at 1000. K and 1200. K from...
Question:
(a) Use a spreadsheet program or graphing software and data at 1000. K and 1200. K from Table 5G.2 to plot the expression for the temperature dependence of ln K given by the dissociation of the diatomic halogens into the atoms, X2 (g) ⇌ 2 X(g).
(b) From the graphs, determine the enthalpies and entropies of dissociation.
(c) Use these data to calculate the standard molar entropies of the gaseous halogen atoms X(g).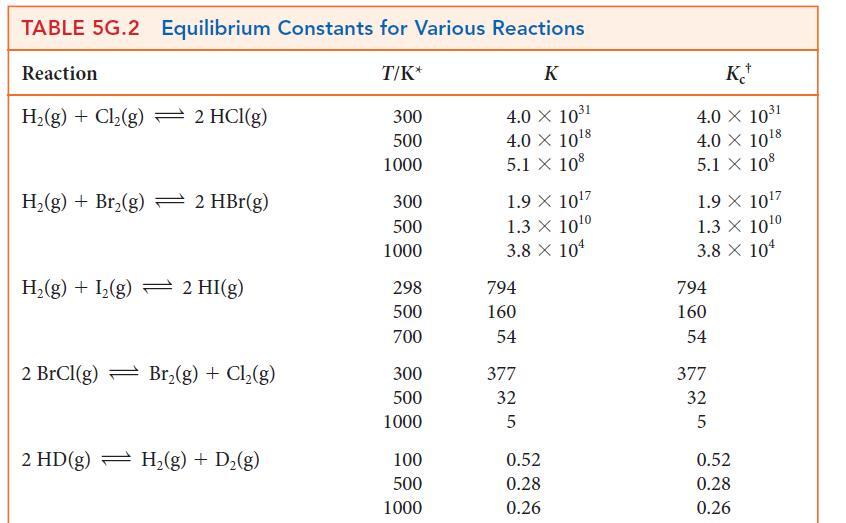
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:





