The van der Waals equation can be arranged into the following cubic equation: (a) Using this equation,
Question:
The van der Waals equation can be arranged into the following cubic equation: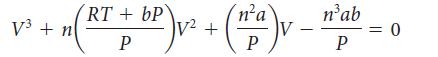
(a) Using this equation, calculate the volume occupied by 0.505 mol NH3 (g) at 25 °C and 95.0 atm. The van der Waals parameters for NH3 are a = 4.225 bar · L2 · mol–2 and b = 3.71 L · mol–2.
(b) Do the attractive or the repulsive forces dominate at this temperature and pressure?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemical Principles The Quest For Insight
ISBN: 9781464183959
7th Edition
Authors: Peter Atkins, Loretta Jones, Leroy Laverman
Question Posted:





