A gas mixture is composed of (15 %) carbon monoxide, (10 %) methane, and the rest air.
Question:
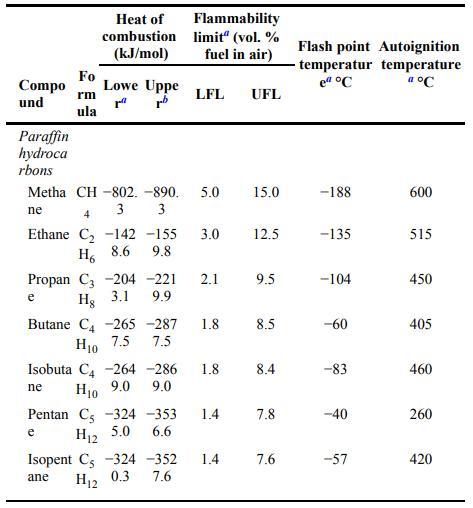
A gas mixture is composed of \(15 \%\) carbon monoxide, \(10 \%\) methane, and the rest air. Is this mixture flammable? Use the flammability data provided in Appendix B.
Data From Appendix B:
Transcribed Image Text:
Compo und Paraffin hydroca rbons Fo rm ula Heat of combustion (kJ/mol) Lowe Uppe rrb Metha CH-802. -890. 5.0 ne 3 3 e Flammability limit" (vol. % fuel in air) 4 Ethane C -142 -155 3.0 9.8 H 8.6 LFL Propan C3 204 -221 2.1 9.9 Hg 3.1 e Butane C4 -265-287 7.5 H10 7.5 Pentan C -324 -353 6.6 H12 5.0 Isopent C -324 -352 7.6 ane H12 0.3 1.8 1.4 UFL Isobuta C4 -264 -286 1.8 8.4 Hio 9.0 9.0 ne 1.4 15.0 12.5 9.5 8.5 7.8 7.6 Flash point Autoignition temperatur temperature el C a C -188 -135 -104 -60 -83 -40 -57 600 515 450 405 460 260 420
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The gas mixture is composed of 15 15 15 carbon monoxide CO and 1...View the full answer

Answered By

Divishad Londhe
I have completed my undergraduate studies as as mechanical engineer and my research focus was Robotics. I have also given GRE and can teach people about prep. I have conducted many robotics workshops in high schools and colleges and taught almost 100-200 students.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemical Process Safety Fundamentals With Applications
ISBN: 9780134857770
4th Edition
Authors: Daniel A. Crowl, Joseph F. Louvar
Question Posted:
Students also viewed these Engineering questions
-
Draw an approximate flammability triangle diagram for methyl alcohol. Use published flammability data from Appendix B and Table 6-3. If a gas containing 20\% methyl alcohol, \(5 \%\) oxygen, and \(75...
-
A gas mixture is found to contain two diatomic A and B species for which the partial pressures of both are 0.05065 MPa (0.5 atm). This mixture is to be enriched in the partial pressure of the A...
-
The financial statements for Leon's are provided in Appendix B at the end of the text. Determine the free cash flow for the year ended December 31, 2011. The $24,999,000 purchase of property, plant,...
-
A car is initially travelling at 3 0 m / s , which is above the speed limit . The driver sees a speed limit trap ahead and applies the brakes for 5 seconds, causing the car to slow down by 2 m / s...
-
Determine the volume and the surface area of the solid obtained by rotating the area of Prob. 5.2 about (a) The x axis, (b) The lines 19 x = in. 9 in 10 in.- 12 In. Sin.
-
Which of the following is true about FUTA obligations? A. FUTA is an employer-only tax B. FUTA is subject to a 5.4% reduction based on employer and state factors. C. FUTA is subject to a $7,000 wage...
-
Calculate the following: 1. \(\left(2.29 \times 10^{3} ight) \times\left(3 \times 10^{4} ight)\) 2. \(\left(6.91 \times 10^{-3} ight) \times\left(9.1 \times 10^{5} ight)\)
-
Zebra Imaginarium, a retail business, had the following cash receipts during December 20--. The sales tax is 6%. Dec. 1 Received payment on account from Michael Anderson, $1,360. 2 Received payment...
-
Salmone Company reported the following purchases and sales ofits only product. Salmone uses a perpetual inventory system.Determine the cost assigned to cost of goods sold using FIFO. DateActivities 2...
-
Estimate the upper and lower flammable limits for carbon monoxide and heptane using the stoichiometric method via Equations 6-10 and 6-11 in the text. Compare to experimental values provided in...
-
What is the flash point temperature \(\left(ight.\) in \(\left.{ }^{\circ} \mathrm{C}ight)\) of a 50 mole percent mixture of methanol and water? Use Appendix B for the flash point temperature for...
-
You are given the following particulars relating to the year ended 31st March, 2016: (a) Total Sales (b) Cost of Goods Sold (c) Expenses for the year (d) The position of current assets and...
-
Consider a cantilevered, rectangular beam subjected to a load pro- duced by a 1 kg point mass at the (free) tip of the beam. Assume that beam material is made of Aluminum and that the beam is 11 in....
-
IntFinMgt 8 1. Explain how a MNC could hedge net receivables in euros with futures contracts. 2. Explain how a MNC could hedge net payables in Japanese yen with futures contracts. 3. Explain how a...
-
An 80km point to point link consists of a 1.5m laser diode with 10mW average output power, followed by a 60km long fibers with 0.2dB/km loss. Assume that dispersion accumulated in fibers are fully...
-
A steel beam is installed in a building when the ambient temperature is - 1 2 C . If the beam if fixed at it s ends ( prevented from expanding / contracting ) , calculate the stress that will develop...
-
In the context of health, communication, innovations, discuss the significance of early adopters within a community. How can health professionals leverage these key individuals to accelerate the...
-
How does a foreign currency option differ from a foreign currency forward contract?
-
Chao, Louis, and Mari, unrelated individuals, own all of the shares of Cerise Corporation. All three shareholders have been active in the management of Cerise since its inception. In the current...
-
Some scientists believe that the collision of one or more large asteroids with the Earth was responsible for the extinction of the dinosaurs. The unit of kiloton is used to describe the energy...
-
A structure known as a cantilever beam is clamped at one end but free at the other, analogous to a diving board that supports a swimmer standing on it (Figure P3.28). Using the following procedure,...
-
Perform measurements as described in Problem P3.28 for cantilever beams of several different lengths. Can you show experimentally that, for a given force F, the deflection of the cantilevers tip is...
-
Family Corporation declared a 30% stock dividend on 400,000 shares of $5 par value common stock outstanding. Market price per common share on this date was $10. What is the amount of retained ea...
-
L Limited acquired an item of equipment on 1 July 2019 for $105000. The estimated useful life of the equipment at the acquisition date was 5 years and the residual value was $5000. The company sold...
-
1. Under 100% sales assumption, using absorption coating, the COGS is calculated by adding Dorect Material (DM), Direct Labor (DL), and Fixed Overhead (FOH) a) True b) False 2. Under 100% sales...

Study smarter with the SolutionInn App


