A plug flow reactor (2 m 3 ) processes an aqueous feed (100 liter/min) containing reactant A
Question:
A plug flow reactor (2 m3) processes an aqueous feed (100 liter/min) containing reactant A (CA0 = 100 mmol/liter). This reaction is reversible and represented by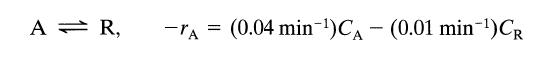
First find the equilibrium conversion and then find the actual conversion of A in the reactor.
Transcribed Image Text:
A = R, -A= (0.04 min-¹)CA - (0.01 min-¹)CR
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To find the equilibrium conversion for the given reversible reaction we need to determine the condit...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
A mixed flow reactor (2 m 3 ) processes an aqueous feed (100 liter/min) containing reactant A (C A0 = 100 mmol/liter). The reaction is reversible and represented by What is the equilibrium conversion...
-
Aqueous feed containing reactant A (C A0 = 2 mol/liter) enters a plug flow reactor (10 liter) which has a provision for recycling a portion of the flowing stream. The reaction kinetics and...
-
Aqueous feed containing reactant A (C A0 = 2 mol/liter) enters a plug flow reactor (10 liter) which has a provision for recycling a portion of the flowing stream. The reaction kinetics and...
-
Assume a retailer has fixed costs of $10,000, a unitvariable cost of $25, and a 50% retail margin. How many units must be sold for her tobreak-even? If she has a target profit of $200,000, how many...
-
For Blue Nile, Inc., what are the expected proceeds to the company? Is this certain? What assumptions are you making? How would you verify this? Amazon.com issued an initial public offering in May...
-
Using the following information, compute cash paid for income taxes. Reported income tax expense . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . $20,000 End of Year Beginning of...
-
Use the exponent distributive rule to expand \((2 \times 19)^{14}\).
-
Answer the following questions related to Dubois Inc. (a) Dubois Inc. has $600,000 to invest. The company is trying to decide between two alternative uses of the funds. One alternative provides...
-
EX 7-15 Lower-of-cost-or-market inventory OBJ. 6 On the basis of the following data, determine the value of the inventory at the lower nc cost or market. Assemble the data in the form illustrated in...
-
The off gas from a boiling water nuclear power reactor contains a whole variety of radioactive trash, one of the most troublesome being Xe-133 (half life = 5.2 days). This off gas flows continuously...
-
An aqueous feed of A and B (400 liter/min, 100 mmol A/liter, 200 mmol B/liter) is to be converted to product in a plug flow reactor. The kinetics of the reaction is represented by Find the volume of...
-
Why would you use a fixed alternative question format in a survey?
-
Reward responses tend to be more intense and longer-lasting than threat responses. Making the case for the change initiative is associated with which organizational change phase? A. Mobilization B....
-
Which among coming up next is right for the class characterized underneath? class understudy { int marks; public: student(){} student(int x) { marks=x; } }; primary() { understudy s1(100);...
-
1. A rock is tossed straight up with a speed of 26 m/s. When it returns, it falls into a hole 14 m deep. Ignore air resistance a. How long the rock in the air, from the instant it is released until...
-
How do structural inequalities in health care and other public services reflect broader patterns of social stratification ?
-
Presented below are selected transactions of Federer AG . Federer sells in large quantities to other companies and also sells its product in a small retail outlet. March 1 Sold merchandise on account...
-
What is meant by reading a defendant his Miranda rights, and why are these rights important?
-
Modify the CYK algorithm so that it applies to any CFG, not just those in CNF.
-
You have two CSTRs and two PFRs, each with a volume of 1.6 m3. Use Figure 2-2(b) to calculate the conversion for each of the reactors in the following arrangements. 1. Two CSTRs in series. 2. Two...
-
The exothermic reaction of stillbene (A) to form the economically important trospophene (B) and methane(C), that is, A B+ C was carried out adiabatically and the following data recorded: The...
-
The financially important reaction to produce the valuable product B (not the real name) was carried out in Jesse Pinkmans garage (see Breaking Bad Season 3, Episode 7). This Breaking Bad,...
-
AirQual Test Corporation provides on-site air quality testing services. The company has provided the following cost formulas and actual results for the month of February: Fixed Component per Month....
-
The monthly salaries for December and the year-to-date earnings of the employees of Bush Consulting Company as of November 30 follow. Determine the amount of social security tax to be withheld from...
-
Kathy Burnett works for Triumph Industries. Her pay rate is $13.32 per hour and she receives overtime pay at one and one-half times her regular hourly rate for any hours worked beyond 40 in a week....

Study smarter with the SolutionInn App


