A mixed flow reactor (2 m 3 ) processes an aqueous feed (100 liter/min) containing reactant A
Question:
A mixed flow reactor (2 m3) processes an aqueous feed (100 liter/min) containing reactant A (CA0 = 100 mmol/liter). The reaction is reversible and represented by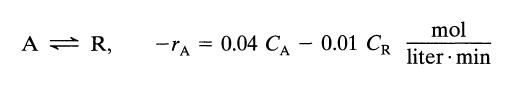
What is the equilibrium conversion and the actual conversion in the reactor?
Transcribed Image Text:
A = R, -TA 0.04 CA - 0.01 CR mol liter min
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
To determine the equilibrium conversion and actual conversion in the mixed flow reactor we need to consider the reversible reaction and use the given ...View the full answer

Answered By

Sarah Khan
My core expertise are:
-_ Finance
-_ Business
-_ Management
-_ Marketing Management
-_ Financial Management
-_ Corporate Finance
-_ HRM etc...
I have 7+ years of experience as an online tutor. I have hands-on experience in handling:
-_ Academic Papers
-_ Research Paper
-_ Dissertation Paper
-_ Case study analysis
-_ Research Proposals
-_ Business Plan
-_ Complexed financial calculations in excel
-_ Home Work Assistance
-_ PPT
-_ Thesis Paper
-_ Capstone Papers
-_ Essay Writing etc...
5.00+
91+ Reviews
92+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Your supervisor has just finished taking a college course in which she learned about flowcharting and data-flow diagrams. In the data-flow diagrams, there are four basic symbols that are used. Write...
-
A plug flow reactor (2 m 3 ) processes an aqueous feed (100 liter/min) containing reactant A (C A0 = 100 mmol/liter). This reaction is reversible and represented by First find the equilibrium...
-
How should we operate a mixed flow reactor so as to maximize the production of R? Separation and recycle of unused reactant is not practical. When aqueous A and aqueous B (C A0 = C B0 ) are brought...
-
Answer the Multple Choice Questions and the code for problem 6in the end PROBLEM 1: General UNIX 1. What is UNIX? a) an operating system b) a text editor c) programming language d) software program...
-
The limit order book for a security is as follows: The specialist receives the following, in order: Market order to sell 300 shares Limit order to buy 100 shares at 25.38 Limit order to buy 500...
-
The Jack n John Corporation has annual sales of $ 6 0 million ( term of sale 2 / 1 0 , net 3 0 ) . The average collection period is 3 0 days. What is the average investment in account receivables as...
-
Explain how you could use the law of large numbers to show that a coin is unfair by tossing it many times.
-
Equity Investment on July 1, 2010, Selig Company purchased for cash 40% of the outstanding capital stock of Spoor Corporation. Both Selig and Spoor have a December 31 year-end. Spoor Corporation,...
-
Q.3. Customers arrive at an order counter with exponential interarrivals with a mean of 10 minutes; the first customer arrives at time 0. A single clerk accepts and checks their orders and processes...
-
A specific enzyme acts as catalyst in the fermentation of reactant A. At a given enzyme concentration in the aqueous feed stream (25 liter/min) find the volume of plug flow reactor needed for 95%...
-
The off gas from a boiling water nuclear power reactor contains a whole variety of radioactive trash, one of the most troublesome being Xe-133 (half life = 5.2 days). This off gas flows continuously...
-
Isabelle Leclerc is the controller at Camden Pharmaceutical Industries, a public company. She is currently preparing the calculation for basic and diluted earnings per share and the related...
-
* Make a well-researched analysis of the factors leading to the failures in (a) NASAs Apollo 18, 19 and 20 programs, and (b) Ford Motors in India in 2021. * Discuss in your paper various ways in...
-
1. What factors need to be in place for a service like the Dabbawallahs to function effectively? 2. What are the economics of the Dabbawallahs' meal distribution network (i.e., operational model)? Is...
-
The system archetype that might be in control of the case study is growth and under-investment. I chose this because the organization was small and they were trying to grow their organization by...
-
Was the crisis response and communication effective? Compare and contrast alternative digital communication efforts and provide alternative recommendations. Crisis One Staples is an office store...
-
One thing that I found working in mental health is the staff generally doesn't accept that the patient's reality is real to them. It can ware down on you at times, and you would be less than...
-
Maggie M iller owns Miller Construction Co. The company maintains accounting records for the purposes of exercising control over its construction activities and meeting its reporting obligations...
-
Whats the difference between an ordinary annuity and an annuity due? What type of annuity is shown below? How would you change the time line to show the other type of annuity?
-
Use the data in Problem P11-4A for the following reaction. The elementary, irreversible, organic liquid-phase reaction A + B C is carried out in a flow reactor. An equal molar feed in A and B enters...
-
The elementary reversible liquid-phase reaction takes place in a CSTR with a heat exchanger. Pure A enters the reactor. (a) Derive an expression (or set of expressions) to calculate G(T) as a...
-
QBR Questions Before Reading. Research has shown (J. Exp. Psychol. Learn. Mem. Cogn., 40, 106114 (2014)) that if you ask a question of the material before reading the material you will have greater...
-
Sunrise Coffee Shop, in an effort to streamline its accounting system, has decided to utilize a cash receipts journal. Record the following transactions for the first two weeks in March, total the...
-
You and your spouse wish to give each of your three grandchildren a yearly tax-free gift from your estate. If you give each of your three grandchildren the maximum amount allowed by law without...
-
Reture 6 Es Allegience Insurance Company's management is considering an advertising program that would require an initial expenditure of $191,980 and bring in additional sales over the next five...

Study smarter with the SolutionInn App


