When a concentrated urea solution is stored it slowly condenses to biuret by the following elementary reaction:
Question:
When a concentrated urea solution is stored it slowly condenses to biuret by the following elementary reaction: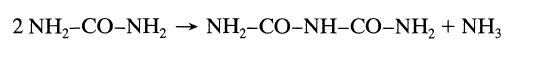
To study the rate of condensation a sample of urea (C = 20 mollliter) is stored at 100°C and after 7 hr 40 min we find that 1 mol% has turned into biuret. Find the rate equation for this condensation reaction. [Data from W. M. Butt, Pak. I. Ch. E., 1, 99 (1973).]
Transcribed Image Text:
2 NH,-CO-NH, NH,-CO-NH-CO–NH, + NH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (3 reviews)
To find the rate equation for the condensation reactionwe can use the following steps Write the rate ...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
A sample containing an alkali sulfate is dried, weighed and dissolved in dilute HCl. Barium chloride solution is added in excess to precipitate barium sulfate, and the precipitate is digested in the...
-
Windy Company manufactures surfboard bags in a one-department process: Design and Manufacturing. The Design and Manufacturing Department uses weighted-average costing method. The following tables...
-
A corporation issues $7,500,000 of 8% bonds to yield interest at the rate of 7%. (a) Was the amount of cash received from the sale of the bonds greater or less than $7,500,000? (b) Identify the...
-
More Info a. Tropical Vista had a January 1, 2024 beginning balance in its Prepaid Rent account of $1,800. During the year the company made payments for prepaid rent of $400. At the end of the year,...
-
34. LO.6 A sculpture that Korliss Kane held for investment was destroyed in a flood. The sculpture was insured, and Korliss had a $60,000 gain from this casualty. He also had a $17,000 loss from an...
-
Daves Restaurant is a chain that employs independent evaluators to visit its restaurants as customers and assess the quality of the service by filling out a questionnaire. The company evaluates...
-
Kindly show calculations and answer fast please. QUESTION 4 [25 MARKS] Yvon Limited manufactures and sells a wide range of machine tools. The company uses absorption costing for both external...
-
The following data are obtained at 0C in a constant-volume batch reactor using pure gaseous A: The stoichiometry of the decomposition is A 2.5R. Find a rate equation which satisfactorily represents...
-
The presence of substance C seems to increase the rate of reaction of A and B, A + B AB. We suspect that C acts catalytically by combining with one of the reactants to form an intermediate, which...
-
Is the population standard deviation known or unknown? The mean speeds of fastball pitches from two different baseball pitchers are to be compared. A sample of 14 fastball pitches is measured from...
-
Sam's grade in a Biology class will be figured on five 1 0 0 . point exams. Sam scored 7 0 % , 8 2 % , 9 1 % , and 6 0 % on the first four exams. What score must Sam score on the fifth exam in order...
-
On January 1, 2024, Adventure World issues $40.4 million of 9% bonds, due in 20 years, with interest payable semiannually on June 30 and December 31 each year. The proceeds will be used to build a...
-
Perform DFS and BFS on the following graph. a b d h g
-
> MyLab Math Course Home | SchoMathway | Algebra Problem Solve X C dadeschools.schoology.com/external_tool/2421768184/launch English 76F Clear Grades Members cos(a)=4/5, a lies in quadrant IV, X...
-
1) out of 115000 students 20% failed in SLC, 40% of the failures failed in Science only and 35% in mathematics only. But 5% of them failed in other subject. Then, i) How many failed in both science...
-
Inland Co., has a total of 40,000 shares of common stock outstanding and no preferred stock. Total stockholders' equity at the end of the current year amounts to $2.5 million and the market value of...
-
Write a while loop that uses an explicit iterator to accomplish the same thing as Exercise 7.3. Exercise 7.3. Write a for-each loop that calls the addInterest method on each BankAccount object in a...
-
Below in Figure P18-11b are two COMSOL simulations for a laminar-flow reactor with heat effects: Run 1 and Run 2. The following figures show the cross section plot of concentration for species A at...
-
Revisit Problem P16-5B. The liquid-phase reaction A B is third order with k 3 = 0.3 dm6/mol2 min and C A0 = 2M. a. What combination of ideal reactors would you use to model the RTD? b. What are the...
-
Revisit Problem P16-3 where the RTD function is a hemicircle. The liquid-phase reaction is first order with k1 = 0.8 min 1 . What is the conversion predicted by a. The tanks-in-series model? b. The...
-
Calla Company produces skateboards that sell for $53 per unit. The company currently has the capacity to produce 95,000 skateboards per year, but is selling 80,700 skateboards per year. Annual costs...
-
Exercise E5-12 15.12 LO 5.4 LO 57 Cost of Goods Sold, Income Statement, and Statement of Compuehensive Income Gaskin Company derne the following items from its adjusted trial balance as of December...
-
can someone please help me with the financial statement to fill in the blanks?! CALCULATOR PRINTER VERSION BACK NE Problem 22-04A -b (Video) Pharoah Company prepares monthly cash budgets. Relevant...

Study smarter with the SolutionInn App


