Given the following equilibrium constants at 427C, Determine the values for the equilibrium constants for the following
Question:
Given the following equilibrium constants at 427°C,
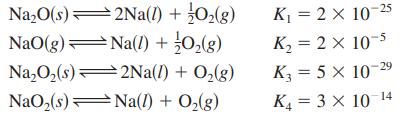
Determine the values for the equilibrium constants for the following reactions:
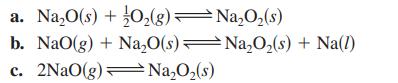
Transcribed Image Text:
2Na(1) + O₂(g) Na₂O(s) NaO(g) Na(1) + O₂(g) Na₂O₂ (s) 2Na(1) + O₂(g) NaO₂(s)Na(l) + O₂(g) K₁ = 2 × 10-25 K₂= 2 × 10-5 K3 = 5 x 10-2 29 K₁ = 3 × 10-14 -
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
To determine the equilibrium constants for the reactions youre ...View the full answer

Answered By

Joseph Ogoma
I have been working as a tutor for the last five years. I always help students to learn and understand concepts that appears challenging to them. I am always available 24/7 and I am a flexible person with the ability to handle a wide range of subjects.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry An Atoms First Approach
ISBN: 9781305079243
2nd Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl
Question Posted:
Students also viewed these Sciences questions
-
Given the following equilibrium constants at 427oC, Na2O(s) 2Na(l) + 12 O2(g) K1 = 2 10-25 NaO(g) Na(l) + l2 O2(g) K2 = 2 10-5 Na2O2(s) 2Na(l) + O2(g) K3 = 5 10-29 NaO2(s) Na(l) + O2(g) K4 = 3...
-
The following equilibrium constants were determined at 1123 K: Write the equilibrium constant expression KP, and calculate the equilibrium constant at 1123 K for C(s) + CO2(g)--2CO(g) CO(g) + Cl2(g)...
-
The following equilibrium constants have been determined for hydrosulfuric acid at 25°C: Calculate the equilibrium constant for the following reaction at the same temperature: H2S(aq) H(aq)HS...
-
What line of code can you add to disable all logging messages in your program?
-
Lazio never reported the income or paid the sales taxes from the Sylvan Beach pizzeria and never obtained workers compensation insurance for its employees. How might these facts have affected the...
-
Assume the current Treasury yield curve shows that the spot rates for 6 months, 1 year, and 1 1/2 years are 1%, 1.1%, and 1.3%, all quoted as semiannually compounded APRs. What is the price of a...
-
Bonds issued at a premium always have a. Interest expense equal to the interest payments b. Interest expense greater than the interest payments c. Interest expense less than the interest payments d....
-
A triply ionized beryllium ion, Be 3+ (a beryllium atom with three electrons removed), behaves very much like a hydrogen atom except that the nuclear charge is four times as great. (a) What is the...
-
Food Sustainable Systems Term Assignment Three: My Sustainable Recipe Grade Value: 30% of Total Grade Due Week 12 Grade Points: (/16) Learning Objective The student will create and prepare either...
-
A chemical constituent flows between three reactors as depicted in Fig. P11.15. Steady-state mass balances can be written for a substance that reacts with first-order kinetics. For example, the mass...
-
For the reaction: K = 1.8 10 -7 at a certain temperature. If at equilibrium [O 2 ] = 0.062 M, calculate the equilibrium O 3 concentration. 30(g) 203(g)
-
At a particular temperature, K = 1.00 10 2 for the reaction In an experiment, 1.00 mole of H 2 , 1.00 mole of I 2 , and 1.00 mole of HI are introduced into a 1.00-L container. Calculate the...
-
What proportion of 15- to 44-year-olds are hypertensive? Hypertension People are classified as hypertensive if their systolic blood pressure (SBP) is higher than a specified level for their age...
-
What are some of the unexpected problems that are likely to come while designing the pattern on propaganda?
-
Can you highlight different applications of the word fairness?
-
Generally speaking, dignity has many shades. What are the shades of dignity discussed in this chapter? Try to find four more shades of dignity.
-
Fairness is a specific word. Do you agree? If yes, give reasons.
-
Write 10 new more challenges following the template used in challenging section.
-
Bill Wheeler established a partnership with June Cramer. The new company, W&C Fuels, purchased coal directly from mining companies and contracted to ship the coal via waterways to a seaport where it...
-
Will the prediction interval always be wider than the estimation interval for the same value of the independent variable? Briefly explain.
-
Pyridine undergoes electrophilic aromatic substitution at the C3 position. Justify this regiochemical outcome by drawing resonance structures of the intermediate produced from attack at C2, at C3,...
-
Predict the product obtained when pyrrole is treated with a mixture of nitric acid and sulfuric acid at 0C.
-
Draw the structure of each of the following compounds: (a) Cyclohexylmethylamine (b) Tricyclobutylamine (c) 2,4-Diethylaniline (d) (1R,2S)-2-Methylcyclohexanamine (e) ortho-Aminobenzaldehyde
-
What is your approach to succession planning and talent development, ensuring continuity of leadership and knowledge transfer within your organization ?
-
Simplify the following expression completely. x+52 - 14 - x+3x-10 Enter the numerator and denominator separately in the boxes below. If the denominator is 1, enter the number 1. Do not leave either...
-
How do you demonstrate resilience as a leader during times of crisis or uncertainty, and what steps do you take to bolster your team's resilience ?

Study smarter with the SolutionInn App


