A voltaic cell is set up utilizing the reaction Under standard conditions, the expected potential is 0.45
Question:
A voltaic cell is set up utilizing the reaction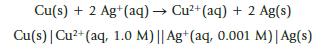
Under standard conditions, the expected potential is 0.45 V. Predict whether the potential for the voltaic cell will be higher, lower, or the same as the standard potential. Verify your prediction by calculating the new cell potential.
Transcribed Image Text:
Cu(s) + 2 Ag+(aq) → Cu²+ (aq) + 2 Ag(s) Cu(s) | Cu²+ (aq, 1.0 M) || Ag+ (aq, 0.001 M)| Ag(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 16% (6 reviews)
The standard potential of a voltaic cell is the potential difference between the cathode and anode a...View the full answer

Answered By

Shehar bano
I have collective experience of more than 7 years in education. my area of specialization includes economics, business, marketing and accounting. During my study period I remained engaged with a business school as a visiting faculty member and did a lot of business research. I am also tutoring and mentoring number of international students and professionals online for the last 7 years.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
A voltaic cell is based on Ag+ (aq) / Ag(s) and Fe3+(aq) / Fe2+ (aq) half-cells. (a) What is the standard emf of the cell? (b) Which reaction occurs at the cathode and which at the anode of the cell?...
-
A voltaic cell is constructed with two Zn2+ - Zn electrodes. The two half-cells have [Zn2+] = 1.8 M and [Zn2+] = 1.00 10-2 M, respectively. (a) Which electrode is the anode of the cell? (b) What is...
-
Telstar Limited was incorporated on 2 January 2015. On 3 January 2015, the company ordered plant from Germany at a cost of E1 million. The plant was loaded free on board in Hamburg on 1 March 2015,...
-
Metro-Goldwyn-Mayer Studios Inc. (MGM) is a major producer and distributor of theatrical and television filmed entertainment. Regarding theatrical films, MGM states, Our feature films are exploited...
-
A mortgage broker is offering a $279,000, 30-year mortgage with a teaser rate. In the first two years of the mortgage, the borrower makes monthly payments on only a 4.5 percent APR interest rate....
-
Describe different views of ethics according to the utilitarian, individual rights, and justice approaches. AppendixLO1
-
On January 1, 2012, Plutonium Corporation acquired 80% of the outstanding stock of Sulfurst Inc. for $268,000 cash. The following balance sheet shows Sulfurst Inc.s book values immediately prior to...
-
Neighborhood Realty, Incorporated, has been operating for three years and is owned by three investors. S. Bhojraj owns 60 percent of the total outstanding stock of 9,020 shares and is the managing...
-
Two half-cells, Pt | Fe 3+ (aq, 0.50 M), Fe 2+ (aq, 1.0 10 5 M) and Hg 2+ (aq, 0.020 M) | Hg, are constructed and then linked together to form a voltaic cell. Which electrode is the anode? What will...
-
The metallurgy of aluminum involves electrolysis of Al 2 O 3 dissolved in molten cryolite (Na 3 AlF 6 ) at about 950C. Aluminum metal is produced at the cathode. Predict the anode product and write...
-
There is some research indicating that college students who use Facebook while studying tend to have lower grades than non-users (Kirschner & Karpinski, 2010). A representative study surveys students...
-
(1) The Mean Value Theorem states: Let f be continuous over the closed [a, b] and differentiable over the open interval (a, b). Then, there exists at least one point c E (a, b) such that: f(b) - f(a)...
-
Assume you are an Israeli investor; the symbol for the Israeli currency, the shekel, is ILS. You see that stock for Top Image has a bid price of ILS 17 and an ask price of ILS 19 in Israel, a bid...
-
3. Given the continuous beam shown below, which span or spans should be loaded with a uniform distributed load to produce a maximum moment at support B? (5 points) SPAN 1 SPAN 2 SPAN 3 A B D 20 ft...
-
Complete the following writing assignment: Analyze the attached 10_pages. Write_about them, summarize what you read, and connect it to personal experiences. CHAPTER 8 Anxiety Disorders DAVID P....
-
As a manager of an airline company you want to learn the average weight of luggages checked in on a flight. From a sample of 1 6 luggages, you find the average to be 2 6 kg and the standard deviation...
-
Find the eigenvalues of A9 for 18 42 7300 312 00 1000
-
Critical reading SAT scores are distributed as N(500, 100). a. Find the SAT score at the 75th percentile. b. Find the SAT score at the 25th percentile. c. Find the interquartile range for SAT scores....
-
What is wrong with the following code for a half adder that must add if add signal equals 1? always @(x) begin if (add == 1) begin sum = x ^ y; carry = x & y; end else begin sum = 0; carry = 0; end...
-
For the following Verilog code, assume that D changes to 1 at time 5 ns. Give the values of A, B, C, D, E, and F each time a change occurs. That is, give the values at time 5 ns, 5 + D, 5 + 2D, and...
-
Assuming B is driven by the simulator command: force B 0 0, 1 10, 0 15, 1 20, 0 30, 1 35 Draw a timing diagram illustrating A, B, and C if the following concurrent statements are executed: always...
-
Ron (78) is single and retired many years ago. during the tax year, he received a federally taxable private pension distribution of $19563. His Ohio income tax base is $55194. What is the amount of...
-
On January 1, 2020, Victory Co. acquired 60% of the shares of Sauce Ltd. by issuing shares valued at $1,500,000. On this date, Sauce Ltd.'s building and machinery had remaining useful lives of 10...
-
Anderson Co. issued a $59,335, 60-day, discounted note to National Bank. The discount rate is 7%. At maturity, assuming a 360-day year, the borrower will pay a.$60,027 b.$59,335 c.$55,182 d.$58,643

Study smarter with the SolutionInn App


