Balance the following equations involving organic compounds. (a) Ag*(aq) + CH;CHO(aq) Ag(s) + C6H5COH(aq) (b) CHCHOH +
Question:
Balance the following equations involving organic compounds.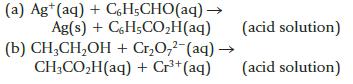
Transcribed Image Text:
(a) Ag*(aq) + CH;CHO(aq)— Ag(s) + C6H5CO₂H(aq) (b) CH₂CH₂OH + Cr₂O,2 (aq) → CH3CO₂H(aq) + Cr³+ (aq) (acid solution) (acid solution) 1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
Balanced equations a Ag aq C6H5CHOaq AgC6H5O2s CH3CO2Haq acid solution b CH3CH2OH Cr2O72aq CH2CO2Haq ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
Pecunious Products, Inc.'s financial results for the past three years are summarized below: Year 3 Year 2 Year 1 Sales trend 179.2 161 140 Current ratio 3.5 3.22 3.08 Acid-test ratio 1.12 1.26 1.54...
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) Ba(OH)-(aq) + HPO 4 (aq )- HCIO4 (aq) + Mg(OH )2 (s)
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) CH3COOH (aq) + KOH(aq)- .co.(aq) + NaO H (aq) - HNO3(aq ) + Ba(OH)2(aq )-
-
In the diagram, the positive terminal of the 12 V battery is grounded it is at zero potential. At what potential is point X? 12 V 4 V Ground
-
Twelve states, some local governments, and private organizations sued the EPA, contending that it did not live up to its obligation under the Clean Air Act to regulate greenhouse gases that result...
-
Should a loan officer ever say no to a business firm requesting a loan? Please explain when and where.
-
On December 31, 2017, James, Inc., borrowed \(\$ 300,000\) on a six percent, 20 -year mortgage note payable. The note is to be repaid in equal semiannual installments of \(\$ 12,979\) (beginning July...
-
Anstead Co. is experiencing a decrease in sales and operating income for the fiscal year ending October 31, 2014. Ryan Frazier, controller of Anstead Co., has suggested that all orders received...
-
Dog Up! Franks is looking at a new sausage system with an installed cost of $695,000. This cost will be depreciated straight-line to zero over the project's 5-year life, at the end of which the...
-
A voltaic cell is constructed in which one half-cell consists of a silver wire in an aqueous solution of AgNO 3 . The other half-cell consists of an inert platinum wire in an aqueous solution...
-
Write balanced equations for the following reduction half-reactions involving organic compounds. (a) HCOH CHO (b) C6H5COH C6HCH3 (c) CH,CH,CHO (d) CH3OH CH CHCHCHOH (acid solution) (acid solution)...
-
How could 1H NMR distinguish between the compounds in each of the following pairs? a. CH3CH2CH2OCH3 and CH3CH2OCH2CH3 b. BrCH2CH2CH2Br and BrCH2CH2CH2NO2 c. d. e. f. g. h. i. CH3 CH, CHa CH3CH CHC...
-
Given the fixed beam shown in the figure below (Fig Q10) solve the following problem. Using three (3) elements of equal length calculate the displacement w, given the following parameters. 1. The...
-
1. Explain how silence can be used effectively in counselling. 2. Building Self-Awareness: Cultural Iceberg Activity Steps for Activity: 1) Find PDF of Iceberg Activity in Week 4 Folder 2) Consider...
-
Success of COVID-19 Vaccination depends on the efficiency of Supply Chain. How? & Why? Elaborate
-
A firm is considering four possible independent project options. Project A has an initial investment cost (in year 0) of $9,000 and yields net benefits of $4,280 each year for 3 years (year 1 to year...
-
what are the key performance indicators of marketing in airline industry ?What is the role of marketing performance measures and financial analysis in organisational performance
-
A poll of 702 frequent and occasional fliers found that 442 respondents favored a ban on cell phones in fight, even if technology permits it. At = .05, can we conclude that more than half the...
-
Maria Castigliani is head of the purchasing department of Ambrosiana Merceti, a medium-sized construction company. One morning she walked into the office and said, The main problem in this office is...
-
Quasimonochromatic light having an irradiance of 400 W/m 2 is incident normally on the cornea (n c = 1.376) of the human eye. If the person is swimming under the water (n w = 1.33), determine the...
-
Compare the amplitude reflection coefficients for an airwater (n w = 4/3) interface with that of an aircrownglass (n g = 3/2) interface, both at near-normal incidence. What are the corresponding...
-
Use Eq. (4.42) and the power series expansion of the sine function to establish that at near-normal incidence we can obtain a better approximation than the one in Problem 4.45, which is [-r ¥ ]...
-
Read (15) ZZZZ Best and 17 Closing the gap 1. Describe the procedures of Arthur Young's partner Larry Gray performed to verify the existence of ZZZZ Best's restoration projects. Were these audit...
-
Perform a search for up-to-date operational and financial information on WGC.Explain and justify your position to the following question utilizing the up-to-date financial data you found in your...
-
5 . To start up a business its founders must invest as follows: Year 0 1 2 3 4 Investment $ 4 , 0 0 0 , 8 0 0 $ 2 , 0 0 0 , 4 0 0 $ 1 , 0 0 0 , 2 0 0 $ 5 0 0 , 1 0 0 $ 2 5 0 , 0 5 0 Following the...

Study smarter with the SolutionInn App


