In the discussion on the composition of air, mention is made of the fact that water vapor
Question:
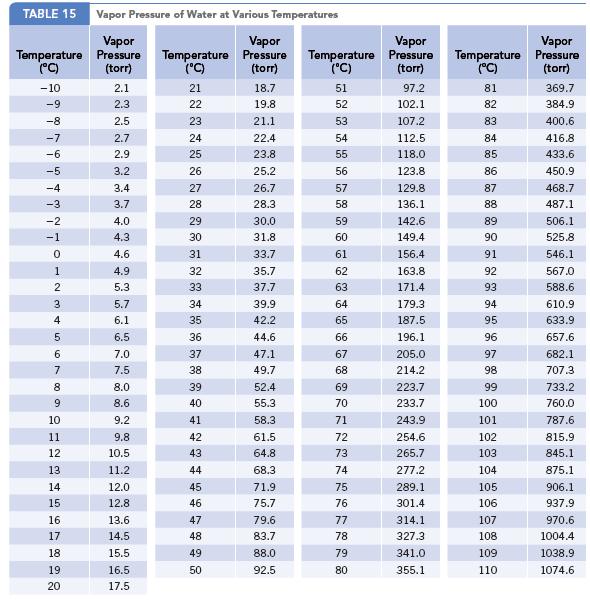
In the discussion on the composition of air, mention is made of the fact that water vapor may have a concentration as high as 40,000 ppm. Calculate the partial pressure exerted by water vapor at this concentration. Assume that this represents a situation with 100% humidity. What temperature would be needed to achieve this value? (See Appendix G.)
Data given in Appendix G
Transcribed Image Text:
TABLE 15 Vapor Pressure of Water at Various Temperatures Vapor Pressure (torr) Temperature (°C) -10 -9 -8 -7 -6 -5 -4 -3 -2 -1 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 2.1 2.3 2.5 2.7 2.9 3.2 3.4 3.7 4.0 4.3 4.6 4.9 5.3 5.7 6.1 6.5 7.0 7.5 8.0 8.6 9.2 9.8 10.5 11.2 12.0 12.8 13.6 14.5 15.5 16.5 17.5 Temperature (°C) 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36 37 38 39 40 41 42 43 44 45 46 47 48 49 50 Vapor Pressure (torr) 18.7 19.8 21.1 22.4 23.8 25.2 26.7 28.3 30.0 31.8 33.7 35.7 37.7 39.9 42.2 44.6 47.1 49.7 52.4 55.3 58.3 61.5 64.8 68.3 71.9 75.7 79.6 83.7 88.0 92.5 Temperature (°C) 51 52 53 54 55 56 57 58 59 60 61 62 63 64 65 66 67 68 69 70 71 72 73 74 75 76 77 78 79 80 Vapor Pressure (torr) 97.2 102.1 107.2 112.5 118.0 123.8 129.8 136.1 142.6 149.4 156.4 163.8 171.4 179.3 187.5 196.1 205.0 214.2 223.7 233.7 243.9 254.6 265.7 277.2 289.1 301.4 314.1 327.3 341.0 355.1 Temperature (°C) 81 82 83 84 85 86 87 88 89 90 91 92 93 94 95 96 97 98 99 100 101 102 103 104 105 106 107 108. 109 110 Vapor Pressure (torr) 369.7 384.9 400.6 416.8 433.6 450.9 468.7 487.1 506.1 525.8 546.1 567.0 588.6 610.9 633.9 657.6 682.1 707.3 733.2 760.0 787.6 815.9 845.1 875.1 906.1 937.9 970.6 1004.4 1038.9 1074.6
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To calculate the partial pressure of water vapor at a concentration of 40000 ppm we can use the foll...View the full answer

Answered By

David Muchemi
I am a professional academic writer with considerable experience in writing business and economic related papers. I have been writing for my clients who reach out to me personally after being recommended to me by satisfied clients.
I have the English language prowess, no grammatical and spelling errors can be found in my work. I double-check for such mistakes before submitting my papers.
I deliver finished work within the stipulated time and without fail. I am a good researcher on any topic especially those perceived to be tough.
I am ready to work on your papers and ensure you receive the highest quality you are looking for. Please hire me to offer my readily available quality service.
Best regards,
4.60+
27+ Reviews
61+ Question Solved
Related Book For 

Chemistry And Chemical Reactivity
ISBN: 9780357001172
10th Edition
Authors: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Question Posted:
Students also viewed these Sciences questions
-
The first quarter of 2008 had not yet ended and Steve Savage already knew the company would surpass the projected $22 million in revenues for the year. He and the management team had doubled sales...
-
Software the Hard Way Joe is a software developer working for Ace Development Company for the last 10 years. He has no management position with the company other than being a software developer,...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
State Newtons second law of motion. What are the limitations on the use of Newtons second law? Explain.
-
Procter and Gamble sold a detergent, Ace con Blanqueador, in Puerto Rico. It advertised that "Mas blanco no se puede" (Whiter is not possible). Clorox, a bleach maker, sued Procter and Gamble...
-
Why do depository institutions face pledging requirements when they accept government deposits?
-
England, Inc., had the following payroll for March: Required Prepare journal entries on March 31 to record: a. Accrual of the monthly payroll. b. Payment of the net payroll. c. Accrual of employer's...
-
1. Wayne owns a shaved ice stand. He sells 700 shaved ice cups per month at $1.50 each, making the total revenue $1050. Each shaved ice costs Wayne $0.50 and he has fixed cost of $750, making his...
-
18. A bank has issued a six-month, $2 million negotiable CD with a 0.52 percent quoted annual interest rate (iCD, sp). (LG 5-2) a. Calculate the bond equivalent yield and the EAR on the CD. b. How...
-
Which of the following atmospheric gases is present in highest concentration in dry air? (a) N 2 O (b) CH 4 (c) O 3 (d) CO
-
A hydrogen-oxygen fuel cell operates on the simple reaction H 2 (g) + 1/2 O 2 (g) H 2 O() If the cell is designed to produce 1.5 A of current and if the hydrogen is contained in a 1.0-L tank at 200...
-
Suppose you want to travel D miles at a constant speed of (60 + x) mi/hr, where x could be positive or negative. The time in minutes required to travel D miles is T(x) = 60D(60 + x) -1 . Show that...
-
How does business-to-business branding differ from consumer products branding? Why should a business-to-business marketer care about building a strong brand?
-
57 =5 Determine the eigen values and the corresponding eigen vectors of the matrix = 0 4 -1 Hence find the matrix P such that P'AP is diagonal matrix. 28-3
-
The stock of FM430.com is trading at $25 today. Exactly one year ago, the stock was trading at $32.50. (b) Assume instead that the stock paid quarterly dividends of $1.25, $2.45, $0.25, $0.25. The...
-
Pick two of my favorite KPIs from The Very Best Digital Metrics For 15 Different Companies and what tools I used to measure them. I can include any KPI related to the entire customer life cycle, such...
-
What are the trends in the job market for Marketing Communications? How is the Marketing Communications field changing? Is the economy affecting the Marketing Communications job market? How so? How...
-
At Ajax Spring Water, a half-liter bottle of soft drink is supposed to contain a mean of 520 ml. The filling process follows a normal distribution with a known process standard deviation of 4 ml. (a)...
-
Record the following selected transactions for March in a two-column journal, identifying each entry by letter: (a) Received $10,000 from Shirley Knowles, owner. (b) Purchased equipment for $35,000,...
-
A 500-nm lightwave in vacuum enters a glass plate of index 1.60 and propagates perpendicularly across it. How many waves span the glass if its 1.00 cm thick?
-
Yellow light from a sodium lamp ( 0 = 589 nm) traverses a tank of glycerin (of index 1.47), which is 20.0 m long, in a time t 1 . If it takes a time t 2 for the light to pass through the same tank...
-
A lightwave travels from point A to point B in vacuum. Suppose we introduce into its path a flat glass plate (n g = 1.50) of thickness L = 1.00 mm. If the vacuum wavelength is 500 nm, how many waves...
-
The Gem (TG) is an organic juice bar located in Philadelphia. TG is particularly famous for its protein smoothies and has two smoothie makers (each is operated by a single employee) customers arrive...
-
GS Enterprises has the following loss distribution for its product liability expenses.If an insurer offered GS a policy with a $100,000 annual deductible, what is the expected claim cost on this...
-
Assume that today is December 3 1 , 2 0 1 9 , and that the following information applies to Abner Airlines: After - tax operating income [ EBIT ( 1 - T ) ] for 2 0 2 0 is expected to be $ 6 5 0...

Study smarter with the SolutionInn App


