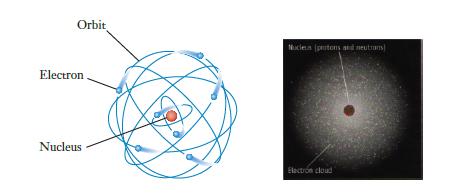
In a typical illustration of the atom such as Figure 2.3, which features lead to misunderstandings about
Question:
In a typical illustration of the atom such as Figure 2.3, which features lead to misunderstandings about the structure of atoms? Which ones give important insight?
Figure 2.3
Transcribed Image Text:
Orbit Electron Nucleus Nocleta (ptons and neutrons) Electron cloud
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
The image provided appears to show two models of the atom On the left there is a depiction of the Bohr model of the atom which presents electrons as p...View the full answer

Answered By

Deborah Joseph
My experience has a tutor has helped me with learning and relearning. You learn everyday actually and there are changes that are made to the curriculum every time so being a tutor has helped in keeping me updated about the present curriculum and all.
I have also been able to help over 100 students achieve better grades particularly in the categories of Math and Biology both in their internal examinations and external examinations.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Chemistry For Engineering Students
ISBN: 9780357026991
4th Edition
Authors: Lawrence S. Brown, Tom Holme
Question Posted:
Students also viewed these Sciences questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
What is the effect of cultural commoditization and transformation on local traditions and customs? Give an example. Check Chapter 4 in the textbook for information on cultural commoditization and...
-
THIRD AVENUE SOFTWARE HEALTH-CARE APP PROJECT This case is new for the ninth edition of Information Technology Project Management . The case provides an opportunity to apply agile and Scrum...
-
Gothic Kings Ltd. Is a 100% owned subsidiary of Hadrian Inc. Gothic has been profitable in the past but incurred a loss for the year ended December 31, 20X3. Hadrian has indicated that if Gothic...
-
Refer to the financial statements and related notes of the Coca-Cola Company in Appendix A of this book. Required 1. Which depreciation method does the company use? Why do you think the company...
-
Consider an automobile dealership with three locations. Data fields exist for stock number, vehicle identification number, make, model, year, color, and invoice cost. Identify the possible candidate...
-
Identify at least three grounds a party may have for objecting to interrogatories.
-
Presented here are liability items for Desmond Inc. at December 31, 2014. Pre-pare the liabilities section of Desmonds balancesheet. 7,800 40,000 80,000 3,500 1,700 Accounts payable Notes payable...
-
Find all the zeros AND factor f into linear factors. 3) f(x) x4 12x2 - 64 = Form a polynomial f(x) with real coefficients having the given degree and zeros. 4) Degree 3: zeros: 1+ and -8 Solve the...
-
Why is the number of protons called the atomic number?
-
One application of conductive polymers is in photovoltaic solar cells. Such devices have traditionally been silicon based. What possible advantages might conduct polymers offer?
-
In Exercise, a) Use the compound interest formula to compute the total amount accumulated b) Determine the interest earned. Round all answers to the nearest cent. $1500 for 4 years at 1.2% compounded...
-
Read Exculpatory Clause and the facts of Buzz Buff v. Awesome Abs, Inc., and formulate a response to various criteria about an exculpatory clause. There is more than one basis for determining whether...
-
Bonnie and Asha, two college students, are in a restaurant. They get into a heated argument about their criminal law examination. Bonnie sweeps her arm across the table angrily, purposely knocking...
-
Company B has entered into a forward rate agreement (FRA) where it will receive 4% with quarterly compounding (for 3-month period) and pay the 3-month LIBOR rate observed in 2 years. The interest...
-
Firm A is a publicly traded company. The equity beta for the company is 1.2, the risk-free rate is 2%, and the market risk premium is 7%. What is the cost of equity for this company?
-
The figure 1 below shows a Third orthographic projection of a drawing, use this drawing to construct an isometric drawing of the figure by making point A the lowest point in your drawing. A 30 15 75...
-
The following data are taken from the comparative balance sheets of Despina Company. Compute and interpret its accounts receivable turnover for year 2011 (competitors average a turnover of 7.5). 2010...
-
Why are stocks usually more risky than bonds?
-
Using acetylene and ethylene oxide as your only sources of carbon atoms, propose a synthesis for each of the following compounds. a. b.
-
Fill in the missing reagents below. Me OEt ?| Br Me SH OEt OMe OEt CN
-
Fill in the missing products below. excess HI Heat 1) Hg(OAc), Et 2) NABH, -: Na MCPBA 1) NaSH 2) H20 HBr
-
Amy runs a wholesale grocery business aiming to provide ingredients to local restaurants and hotels in Hunter Region area, and she hires an accountant to record the transactions for her business....
-
Using the table below, calculate the menu mix percent data and the number of each dish likely to sell based on a forecast of 482 guests, if only 71% of guests order appetizers Menu Item Number Sold...
-
It costs a total of $2,000 to perform preventive maintenance (PM) on five identical production machines. This is the cost to perform preventive maintenance on all five of the machines.If one of the...

Study smarter with the SolutionInn App


