Calculate the mass defect of 4 He. The atomic mass of the atom is 4.002602 atomic mass
Question:
Calculate the mass defect of 4He. The atomic mass of the atom is 4.002602 atomic mass units (u) .
Strategy Use the information in Table 21.3, to calculate the atomic masses of the nucleons; then calculate the mass defect by the difference from its atomic mass of 4.002602 u.
Table 21.3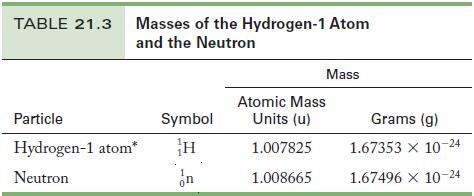
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:





