Write the half-reaction and the chemical equations for the reactions that occur in the electrolysis of (a)
Question:
Write the half-reaction and the chemical equations for the reactions that occur in the electrolysis of
(a) Molten CaCl2, using inert electrodes.
(b) A saturated solution of magnesium sulfate, using inert electrodes.
(c) The electrolysis cell represented in this diagram.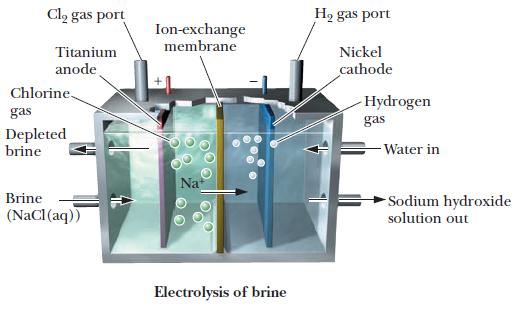
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
a A...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry Principles And Practice
ISBN: 9780534420123
3rd Edition
Authors: Daniel L. Reger, Scott R. Goode, David W. Ball
Question Posted:
Students also viewed these Sciences questions
-
Sharp Company manufactures a product with the following standards: Direct materials Direct labor Standard Quantity or Hours 3 feet ? hours Standard Price or Rate Standard Cost $5 per foot ? per hour...
-
Write the half-reactions and the balanced chemical equations for the reactions that occur in the electrolysis of (a) A zinc chloride aqueous solution, using zinc electrodes. (b) A calcium bromide...
-
Write balanced net ionic equations for the reactions that occur in each of the following cases. Identify the spectator ion or ions in each reaction. (a) Cr2 (SO4)3(aq) + (NH4)2CO3(aq) (b) Ba...
-
1. [20 points] A mattress manufacturer has three production facilities located in Pittsburg (PA), Houston (TX), and Los Angeles (CA). They supply their products to three distribution centers in...
-
Consider two diffuse surfaces A 1 and A 2 on the inside of a spherical enclosure of radius R. Using the following methods derive an expression for the view factor F 12 in terms of A 2 and R. (a) Find...
-
The Ogden Corporation makes an investment of $25,000, which yields the following cash flows: a. What is the present value with a 9 percent discount rate (cost of capital)? b. What is the internal...
-
Write the following in standard form. a. \((4+5 i)(2-3 i)\). b. \((1+i)^{3}\). c. \(\frac{5+3 i}{1-i}\).
-
Waterways has two major public-park projects to provide with comprehensive irrigation in one of its service locations this month. Job J57 and Job K52 involve 15 acres of landscaped terrain which will...
-
. Question 12 Dabney Software Co. is trying to estimate its optimal capital structure. Right now, Simon has a capital structure that consists of 20% debt and 80% equity, based on market values....
-
A solution contains the ions H + , Ag + , Pb 2 + , and Ba 2 + , each at a concentration of 1.0 M. (a) Which of these ions would be reduced first at the cathode during an electrolysis? (b) After the...
-
A possible reaction for a fuel cell is (a) Write the oxidation and reduction half-reactions that occur, assuming a basic electrolyte. (b) Use standard free energies of formation from Appendix G to...
-
The parametric equations where - u and - v represent the surface shown below. Try to create your own parametric surface using a computer algebra system. x = 3 + sin u[7 - cos(3u2v) - 2 cos(3u...
-
Terry recently read an article about portfolio diversification possibilities and the Sharpe ratio. Currently, he owns an optimal risky portfolio. After reading the article he decides to incorporate...
-
Discuss changes to the social security system that were made during 2020. Discuss the potential range of future changes to social security. Discuss how potential future changes might affect Jill and...
-
aramysh/Shutterstock a. Open w05p1 Build and save it as w05p1Build LastFirst. Show nonprinting characters if they are not already displayed. b. Ensure that the insertion point is at the beginning of...
-
you buy a bond for $2,500 and it pays 2% annual interest for 10 years. How much would you receive in interest payments each year? After how much time would you get back your initial investment of...
-
In the United States, federal taxes on investors' capital gains income are lower than the tax rates on dividend income. Despite being taxed at a higher rate than capital gains, why would a...
-
On December 31, 2014, PanTech Company invests $20,000 in SoftPlus, a variable interest entity. In contractual agreements completed on that date, PanTech established itself as the primary beneficiary...
-
At the beginning of the year, Lam Ltd. had total assets of $800,000 and total liabilities of $500,000. Use this information to answer each of the following independent questions. (a) If Lam's total...
-
A light foam cup similar to a disposable coffee cup has a weight of 0.05 N. A steel bar is placed inside the cup. The bar has a specific weight of 76.8 kN/m 3 , a diameter of 38.0 mm, and a length of...
-
Repeat Problem 5.29, but consider that the steel bar is fastened outside the bottom of the cup instead of being placed inside. Repeat Problem A light foam cup similar to a disposable coffee cup has a...
-
Figure 5.27 shows a raft made of four hollow drums supporting a platform. Each drum weighs 30 lb. How much total weight of the platform and anything placed on it can the raft support when the drums...
-
models in healthcare that apllies to communication and socio - economic influences
-
ezto.mheducation.com/ext/map/index.html?_con=con&external_browser=0&launchUrl=https%253A%252F%252Fnewconnect.mhe xam 2 (Chapters 6 & 7) 10. Problem (2 parts) 10 2 Dints 00:57:28 Saved You loaned...
-
Research for a bond mutual fund offered here in the Philippines A. Name of theFund B. The composition of the Fund C. When is the Inception Date of the Fund D. Explain the current net asset value per...

Study smarter with the SolutionInn App


