From examination of the molecular models iv, choose the substance that (a) Can be hydrolyzed to form
Question:
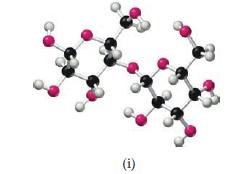
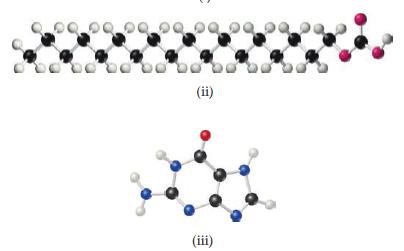
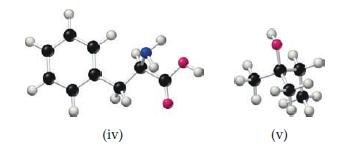
From examination of the molecular models i–v, choose the substance that
(a) Can be hydrolyzed to form a solution containing glucose
(b) Is capable of forming a zwitterion
(c) Is one of the four bases present in DNA
(d) Reacts with an acid to form an ester
(e) Is a lipid.
Transcribed Image Text:
(i)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
a From the structure it looks like this may be a molecule with two glucose units attached to each ot...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 9780321910417
13th Edition
Authors: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
From examination of the molecular models i-v, choose the substance that (a) can be hydrolyzed to form a solution containing glucose, (b) is capable of forming a zwitterion, (c) is one of the four...
-
Waxes can be hydrolyzed to yield an alcohol and a carboxylic acid. Draw the products obtained when triacontyl hexadecanoate undergoes hydrolysis.
-
DNA is capable of forming complex helical structures. An unusual triple-helix structure of poly(dA).2poly(dT) DNA was studied by P. V. Scaria and R. H. Shafer [Journal of Biological Chemistry 266...
-
Top Bun, an international bread empire that reports under IFRS. Their fiscal year-end is December 31, 2022. They began the year on January 1, 2022 with 102116 common shares. They issued 56760 common...
-
Refer to Equations (4.21), (4.22), (4.25), and (4.27). Let x3i = 2x2i. Show why it is impossible to estimate these equations.
-
What lessons can supervisors and organizations learn from this line of applied research?
-
Dispatch the shipment. Orders are packaged, shipping documents prepared, and goods loaded on the right vehicle. LO.1
-
Coca-Cola markets PowerAde as a sports drink that competes with Gatorade. Competition for sports drinks is fierce where they are sold in the coolers of convenience stores. Coca-Cola is thinking about...
-
please help!!!! thank you so much Question 24 Accounting income is synonymous with economic income. A True B False Question 25 The trial balance: A is used to check the equality of debits and credits...
-
Nicholas is the facilities manager for Green Market Groceries. The store is remodeling and wants to determine which brand of freezer to use for its frozen goods section. The freezers vary by size,...
-
The coordination complex [Cr(CO) 6 ] forms colorless, diamagnetic crystals that melt at 90C. (a) What is the oxidation number of chromium in this compound? (b) Given that [Cr(CO) 6 ] is diamagnetic,...
-
Indicate whether each statement is true or false. (a) Butane contains carbons that are sp 2 hybridized. (b) Cyclohexane is another name for benzene. (c) The isopropyl group contains three sp 3...
-
Describe the franchise relationship? Or what does Bagelz have to do to support franchisees and alternately, what does a franchisee bring to the relationship with Bagelz?
-
Before beginning a study investigating the ability of the drug heparin to prevent bronchoconstriction, baseline values of pulmonary function were measured for a sample of 12 individuals with a...
-
which of the following (list all that apply) are advantages of a balanced binary search tree over an unbalanced one: 1. it requires less memory 2. it's faster to move from node to node 3. it's faster...
-
6) Do you find conditional probability problems challenging? Have you tried watching the videos on canvas and has it helped?
-
1. Determine the cost of heating 3 gallons of water (water weighs 8.33L per gallon ) at a room temperature of 22 degrees Celsius to the boiling point of 100 degrees Celsius at the energy rating of...
-
Writer One Inc. manufactures ball point pens that sell at wholesale for $0.80 per unit. Budgeted production in both 2018 and 2019 was 16,000 units. There was no beginning inventory in 2018. The...
-
Dawn started her own rock band on January 2, 2018. She acquired all her equipment on January 2, 2018, and did not dispose of any of it before 2020. On April 15, 2020, the bands amplifiers, speakers,...
-
In a large midwestern university, 30% of the students live in apartments. If 200 students are randomly selected, find the probability that the number of them living in apartments will be between 55...
-
What is the minimum number of atoms that could be contained in the unit cell of an element with a face-centered cubic lattice?
-
The unit cell of nickel arsenide is shown here. (a) What type of lattice does this crystal possess? (b) What is the empirical formula? -3.57 A- ( 120 side view top view Ni As
-
The unit cell of a compound containing strontium, iron, and oxygen is shown here. (a) What type of lattice does this crystal possess (all three lattice vectors are mutually perpendicular)? (b) What...
-
Arnold inc. is considering a proposal to manufacture high end protein bars used as food supplements by body builders. The project requires an upfront investment into equipment of $1.4 million. This...
-
Billy Bob bank has three assets. It has $83 million invested in consumer loans with a 3-year duration, $46 million invested in T-Bonds with a 12-year duration, and $69 million in 6-month (0.5 years)...
-
Ventaz Corp manufactures small windows for back yard sheds. Historically, its demand has ranged from 30 to 50 windows per day with an average of 4646. Alex is one of the production workers and he...

Study smarter with the SolutionInn App


