The first stage of treatment at the reverse osmosis plant in Carlsbad, California, is to flow the
Question:
The first stage of treatment at the reverse osmosis plant in Carlsbad, California, is to flow the water through rock, sand, and gravel as shown here. Would this step remove particulate matter? Would this step remove dissolved salts?
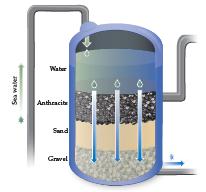
Transcribed Image Text:
Sea water Anthracite Sand Gravel
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
Yes this step would remove partic ulate matter The ...View the full answer

Answered By

Isabel Seraspi
I have experience teaching math, science, and English to students of all ages. I have also worked as a tutor in a college setting, helping students with their homework and preparing them for exams.
I believe that tutoring is a great way to help students learn. It allows students to get one-on-one help with their studies, and it gives them the chance to ask questions and get immediate feedback. Tutoring can also be tailored to the individual needs of the student, which is why I believe it is so effective.
I have seen firsthand how tutoring can help students improve their grades and confidence. I have also seen how it can help students who are struggling with a particular subject. I believe that tutoring is a great way to help students learn and succeed in school.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry The Central Science
ISBN: 978-0134414232
14th Edition
Authors: Theodore Brown, H. LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward, Matthew Stoltzfus
Question Posted:
Students also viewed these Sciences questions
-
The burn time for the first stage of a rocket is a normal random variable with a standard deviation of 0.8 minute. Assume a normal prior distribution for p with a mean of 8 minutes and a standard...
-
The first stage of a Saturn V space vehicle consumed fuel and oxidizer at the rate of 1.50 X 104 kg/s, with an exhaust speed of 2.60 X 103 m/s. (a) Calculate the thrust produced by these engines. (b)...
-
The first stage of an activity-based costing system does not involve: Identifying activities and its attributes Identifying activity attributes Assigning costs Preparing activity dictionary
-
BC Lessee Co. enters a non-cancelable lease agreement with Blackcomb Co. on January 1, 2023, under which Blackcomb Co. leases equipment to BC Lessee Co. Information pertaining to the lease is...
-
Ms. Kona owns a 10 percent interest in Carlton LLC. This year, the LLC generated $72,400 ordinary income. Ms. Konas marginal tax rate is 33 percent, and she does not pay SE tax on her LLC income. a....
-
No meat-eating vegetarians are individuals with a high-protein diet. Therefore, no individuals with a high-protein diet are meat-eating vegetarians. Use either the traditional square of opposition or...
-
What if sales price could be increased by 20 per cent?
-
Misty needs to have $15,000 at the end of 5 years to fulfill her goal of purchasing a small sailboat. She is willing to invest a lump sum today and leave the money untouched for 5 years until it...
-
Finishing Touches has two classes of stock authorized: 8%, $10 par preferred, and $1 par value common. The following transactions affect stockholders' equity during 2021, its first year of...
-
Edison operates a small, fast-growing electronics business. His workload has expanded to the point where he decides to hire a full-time manager. He will then take one year off to travel, and on his...
-
A solution containing several metal ions is treated with dilute HCl; no precipitate forms. The pH is adjusted to about 1, and H 2 S is bubbled through. Again, no precipitate forms. The pH of the...
-
Calculate the solubility of LaF 3 in grams per liter in (a) Pure water, (b) 0.010 M KF solution, (c) 0.050 M LaCl 3 solution.
-
Trim Corporation purchased 75 percent of the voting common stock of Round Corporation on January 1, 20X2, for \(\$ 300,000\). At that date, Round reported the following summarized balance sheet data:...
-
Context This task requires analysing a network scenario, design the network architecture and recommend IT solutions including ethical, security and sustainability considerations.The purpose of this...
-
What was the Prime Cost Percent for Mandy's BBQ Pit for August? Select one: a. 46.5% b. 73.9% c. 63.4% d. 85%
-
Finding Critical Values and Confidence Intervals. In Exercises 5-8, use the given information to find the number of degrees of freedom, the critical values x? and x*, and the confidence interval...
-
An investor sold 100 shares of ABC stock short at $25 and buys one ABC Jan 30 call @ $5. What is this investor's maximum gain, maximum loss, and breakeven points from this strategy?
-
Jake, Sachs and Brianne own a tour company called Adventure Sports. The partners share profits and losses in a 1:3:4 ratio. After Lengthy Dissagreements among the partners and several unprofitable...
-
1. If you were Samantha Parks, how would you prioritize which projects or parts of projects to delegate? 2. In explaining what makes her decisions hard, Parks said, I hire good people, creative...
-
Suppose that the electrical potential at the point (x, y, z) is E(x, y, z) = x + y - 2z. What is the direction of the acceleration at the point (1,3,2)?
-
Determine which of the following statements are false and correct them. (a) The frequency of radiation increases as the wavelength increases. (b) Electromagnetic radiation travels through a vacuum at...
-
Arrange the following kinds of electromagnetic radiation in order of increasing wavelength: infrared, green light, red light, radio waves, X-rays, ultraviolet light.
-
(a) What is the frequency of radiation that has a wavelength of 10m, about the size of a bacterium? (b) What is the wavelength of radiation that has a frequency of 5.50 1014s-1? (c) Would the...
-
Physical Units Method, Relative Sales Value Method Farleigh Petroleum, Inc., is a small company that acquires high - grade crude oil from low - volume production wells owned by individuals and small...
-
A proposed $2.5 M investment in new equipment at a 100 MG/y M&Ms factory will save the plant $800,000/y in energy costs. Assuming an annual interest rate of 5%/y (compounded annually), and an...
-
Brief Exercise 10-7 Coronado Company obtained land by issuing 2,250 shares of its $14 par value common stock. The land was recently appraised at $103,240. The common stock is actively traded at $44...

Study smarter with the SolutionInn App


