Figure P18.74 shows a point charge. At which point (A, B, or C) is the energy density
Question:
Figure P18.74 shows a point charge. At which point (A, B, or C) is the energy density (the energy per unit volume) largest?
Figure P18.74
?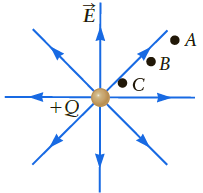
Transcribed Image Text:
A ●B C +Q
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
The electric energy density is proportional to the electric field strength squared So we need to f...View the full answer

Answered By

Gaurav Soni
Teaching was always an area where I can pursue my passion. I used to teach my friends and junior during my school and college life. After completing my professional qualification (chartered accountancy) and before joining my job, I also joined an organization for teaching and guidance to my juniors. I had also written some articles during my internship which later got published. apart from that, I have also given some presentations on certain amendments/complex issues in various forms.
Linkedin profile link:
https://www.linkedin.com/in/gaurav-soni-38067110a
5.00+
7+ Reviews
13+ Question Solved
Related Book For 

College Physics Reasoning and Relationships
ISBN: 978-0840058195
2nd edition
Authors: Nicholas Giordano
Question Posted:
Students also viewed these Sciences questions
-
Exercise 23.43 shows that, outside a spherical shell with uniform surface charge, the potential is the same as if all the charge were concentrated into a point charge at the center of the sphere. (a)...
-
1. Figure 16-50 shows electric field lines due to a point charge. What can you say about the field at point 1 compared with the field at point 2? (a) The field at point 2 is larger, because point 2...
-
Figure P18.40 shows several equipotential surfaces that result from a point charge at the origin. (a) If V 0 = +20 V, is this point charge positive or negative? (b) An external force moves an...
-
Gabriele Enterprises has bonds on the market making annual payments, with seven years to maturity, a par value of $1,000, and selling for $974. At this price, the bonds yield 7.2 percent. What must...
-
Bond J is a 4 percent coupon bond. Bond K is a 12 percent coupon bond. Both bonds have nine years to maturity, make semiannual payments, and have a YTM of 8 percent. If interest rates suddenly rise...
-
Chittenden County had the following federal award activity during the most recent fiscal year: Required a. Based on size, which programs would be considered Type A programs? Type B programs? b. You...
-
Still staying with the data in Example 6.1, let us assume not only that the sales volume has the same three possible outcomes, but that, independent of the sales volume, the cost of labour will be: l...
-
Margaret Cleary is a paralegal who works for a law firm that requires her to bill clients seven hours a day. One of Margarets assignments was to prepare interrogatories on a real estate case. She...
-
The income statement of Whitlock Company is presented here. WHITLOCK COMPANY Income Statement For the Year Ended November 30, 2020 Sales revenue $ 7,504,800 Cost of goods sold Beginning inventory $...
-
Two astronomers in different parts of the world make measurements M1 and M2 of the number of stars N in some small region of the sky, using their telescopes. Normally. There is a small possibility e...
-
A parallel-plate capacitor with C = 10 F is charged so as to contain 1.2 J of energy. If the capacitor has a vacuum between plates that are spaced by 0.30 mm, what is the energy density (the energy...
-
If the charge Q in Figure P18.74 is doubled, by what factor does the energy density change at point A? Figure P18.74 ? A B C +Q
-
Comment on the potential of oil shale and oil sands as future energy sources.
-
The figure shows a turbine-driven pump that provides water, at high pressure, to a tank located 25-m higher than the pump. Steady-state operating data for the turbine and the pump are labelled on the...
-
Step 1 Step 2 1. Sketch what step 4 and then step 5 would look like. Step 4 Step S 2. How many black triangles are in each step? Step 1 black A = | Step 2 = 4 black A's step 3 = 13 black D's 3. What...
-
The pressure cooker pictured here consists of a light pressure vessel with a heavy lid of weight W. When the lid is secured, the vessel is filled with a hot pressurized gas of pressure p. After some...
-
5) A large group of students took a test in Finite Math where the grades had a mean of 72 and a standard deviation of 4. Assume that the distribution of these grades is approximated by a normal...
-
Q9 (5 points) According to Dr. Henry Mintzberg, a noted management scholar from McGill University in Montreal, PQ, "business organizations perform only two activities of consequence." What are these...
-
What is a Fourier integral? A Fourier sine integral? Give simple examples.
-
You are interested in investing and are considering a portfolio comprised of the following two stocks. Their estimated returns under varying market conditions are provided: (note: it is difficult to...
-
To what volume should you dilute 0.300 L of a 6.00 M KOH solution to obtain a 2.50 M KOH solution?
-
What volume (in L) of 2.50 M NaCl Solution is required to completely react with 0.150 L of a 0.150 M AgNO 3 solution according to the reaction: NaCl(aq) + AgNO3(aq) AgCl(s) + NaNO 3 (aq)
-
Write an equation for the precipitation reaction that occurs (if any) when solutions of calcium chloride and potassium carbonate are mixed.
-
QUESTION 3 A business owns seven flats rented out to staff at R500 per month. All flats were tenanted Ist january 21 months rent was in arrears and as at 31st December 14 months' rent wa Identify the...
-
1. 2. 3. Select the Tables sheet, select cells A6:B10, and create range names using the Create from Selection button [Formulas tab, Defined Names group]. Select cells B1:F2 and click the Name box....
-
Tropical Rainwear issues 3,000 shares of its $18 par value preferred stock for cash at $20 per share. Record the issuance of the preferred shares. (If no entry is required for a particular...

Study smarter with the SolutionInn App


