Question: Identify the following functional groups in this organic molecule: amide, ester, ketone, ether, alcohol, aldehyde, amine. C CH CH I CH, O CH H3CO. CH
Identify the following functional groups in this organic molecule: amide, ester, ketone, ether, alcohol, aldehyde, amine.
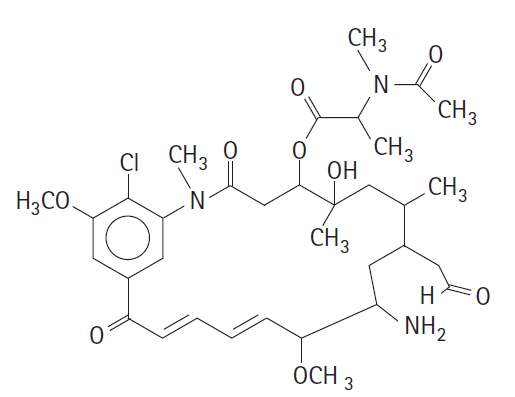
C "CH CH I CH, O CH H3CO. CH * NH2
Step by Step Solution
3.52 Rating (152 Votes )
There are 3 Steps involved in it
1 Ether 2 Amide 3 Ester 4 Ami... View full answer

Get step-by-step solutions from verified subject matter experts


