The yield of a chemical process was measured using five batches of raw material, five acid concentrations,
Question:
The yield of a chemical process was measured using five batches of raw material, five acid concentrations, five standing times (A, B, C, D, E), and five catalyst concentrations (α, β, γ, δ, ∈). The Graeco-Latin square that follows was used. Analyze the data from this experiment (use α = 0.05) and draw conclusions.
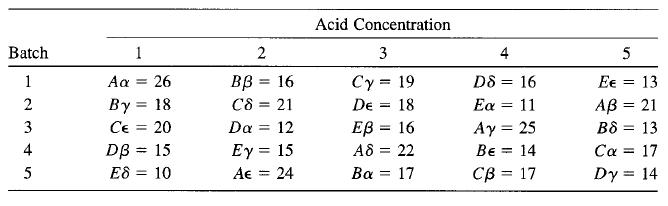
Transcribed Image Text:
Acid Concentration Batch 1 2 4 5 D8 = 16 Εα 11 Ay = 25 1 26 вв 3 16 Cy = 19 Aa Ee = 13 %3D By = 18 Ce = 20 DB = 15 E8 = 10 2 C8 21 De 18 AB = 21 %3D %3D 3 Da = 12 ЕВ 3 16 B8 = 13 %3D Ey = 15 Ae = 24 4 A8 = 22 Be = 14 Ca = 17 %3D %3D 5 Ba = 17 Св 3 17 Dy = 14 %3D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
Batch Acid Time Catalist Yield 1 1 A a 26 2 1 B c 18 3 ...View the full answer

Answered By

Anyango Anyango
Immediately after I finished secondary school waiting to join university I was practising as a tutor of business because of my passion and interests. I have done that for the longest time even though pursuing a different carrier.
I also love playing with figures to solve mathematical problems.
Above all this, I love assisting learners to scale higher and get good grades. Most of the time I would volunteer to teach students in the neighbourhood with their assignments.So far I have seen most of them improve well in their academics and even pass with flying colours and able to join the university.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The yield of a chemical process is being studied. From previous experience yield is known to be normally distributed and = 3. The past five days of plant operation have resulted in the following...
-
A production supervisor at a major chemical company wishes to determine whether a new catalyst, catalyst XA-100, increases the mean hourly yield of a chemical process beyond the current mean hourly...
-
Referring to Exercise 8.21 (page 307), regard the sample of five trial runs (which has standard deviation 19.65) as a preliminary sample. Determine the number of trial runs of the chemical process...
-
93) Clay of the Land is a manufacturer of glazed clay pots. Currently, it produces 300 clay pots per month which it sells through nurseries at a constant price of $5 per pot. Current demand for clay...
-
Find the equilibria of the following discrete-time dynamical system from the graphs of their updating functions Label the coordinates of the equilibria. 10 Time th)
-
3.3 Why is it necessary for a firm to have a number of separate ledger accounts in its books of account?
-
Web-based exercise. A popular saying in golf is You drive for show but you putt for dough. You can find this seasons Professional Golfers Association (PGA) tour statistics at the PGA tour Web site:...
-
Refer to the data in Exercise 9.15. Data in 9.15 Oerstman, Inc., uses a standard costing system and develops its overhead rates from the current annual budget. The budget is based on an expected...
-
helpppp me plz chegg i nedd it now To verify the client's cutoff of cash receipts, the auditors may be on hand to count the undeposited cash receipts on the last business day of the period. True False
-
Victoria Dauzat and Phyllis Jeansonne were shopping at a Dollar General store. Security cameras were in operation at the store that day. A Dollar General employee, Amanda Poarch, commenced...
-
Suppose that in Problem 4-14 the data taken on day 5 were incorrectly analyzed and had to be discarded. Develop an appropriate analysis for the remaining data. Problem 4-14 A chemist wishes to test...
-
Suppose that in Problem 4-15 the engineer suspects that the workplaces used by the four operators may represent an additional source of variation. A fourth factor, workplace (, , , ) may be...
-
Study the following Excel multiple regression output. How many predictors are in this model? How many observations? What is the equation of the regression line? Discuss the strength of the model in...
-
(Reference: A Closer Look on Cost Accounting, De Jesus, 2019) Paulo Corporation had the following account balances as of August 1, 2020: Raw materials inventory (direct and indirect) Work in Process...
-
Use the given data values (a sample of female arm circumferences in centimeters) to identify the corresponding z scores that are used for a normal quantile plot, then identify the coordinates of each...
-
1. What stakeholders other than customers, suppliers, and partners should be considered when conceiving ways to monetize data? 2. What would be one strategy to maximize "reliability" attribute in...
-
What important guidance does little's law offer healthcare leadership regarding the planning and utilization of resources
-
Semester Two Practice Examinations, 2022 STAT2201 Question 2. [10 marks] A study investigated the effect of playing computer games on heart rate. Twenty eight individuals were recruited into the...
-
Refer to Exercise 15. Data From Exercise 15: Mr. James McWhinney, president of Daniel-James Financial Services, believes there is a relationship between the number of client contacts and the dollar...
-
Evaluate each logarithm to four decimal places. log 0.257
-
A 20.0-mL solution of 0.005 00 M Sn 2+ in 1 M HCl was titrated with 0.020 0 M Ce 4+ to give Sn 4+ and Ce 3+ . Calculate the potential (versus S.C.E.) at the following volumes of Ce 4+ : 0.100, 1.00,...
-
Would indigo tetrasulfonate be a suitable redox indicator for the titration of Fe(CN) 6 4- with Tl 3+ in 1 M HCl? The potential at the equivalence point must be between the potentials for each redox...
-
Compute the titration curve for Demonstration 15-1, in which 400.0 mL of 3.75 mM Fe 2+ are titrated with 20.0 mM MnO 4 - at a fixed pH of 0.00 in 1 M H 2 SO 4 . Calculate the potential versus S.C.E....
-
Randy (48) takes a $22,000 distribution from his IRA to pay some of his daughter's $28,000 qualified education expenses at an eligible educational institution. His daughter paid $18,000 of her...
-
The takeover specialist chose to use the value derived from dividend discount model, while the directors prefer to use Net Realisable Value approach. Critically discuss the reasoning of each parties...
-
Tony and Suzie graduate from college in May 2021 and begin developing their new business. They begin by offering clinics for basic outdoor activities such as mountain biking or kayaking. Upon...

Study smarter with the SolutionInn App


