Show that the components of the C ij matrix in Eq. (4.2.2) are related to the components
Question:
Show that the components of the Cij matrix in Eq. (4.2.2) are related to the components of Cijkl by the relation:

Equation 4.2.2
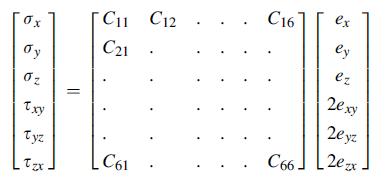
Transcribed Image Text:
Cij = C1 C1122 C1133 C1112 C1123 C1131 C2211 C2222 C2233 C2212 C2223 C2231 C3311 C3322 C3333 C3312 C3323 C3331 C1211 C1222 C1233 C1212 C1223 C1231 C2311 C2322 C2333 C2312 C2323 C2331 C3111 C3122 C3133 C3112 C3123 C3131.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
From relation 421 0 C C3e 2042C150 2C16ex 0 Ce C230 2C246 2C25 2C260x O C31 ...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Elasticity Theory Applications And Numerics
ISBN: 9780128159873
4th Edition
Authors: Martin H. Sadd Ph.D.
Question Posted:
Students also viewed these Engineering questions
-
Show from Figure 11-9C that the components of w along the fixed (xi) axes are wi = 0 cos o + sin Osin o wi = sin o sin 0 cos o wj = j cos 0 +
-
Let j (r) be an arbitrary current distribution. (a) Show that the components of the magnetic dipole moment m = 1/ 2 d 3 r r j are invariant to a rigid shift of the origin of coordinates. (b) Show...
-
The following table lists the components needed to assemble an end item, the manufacturing or purchase lead times (in weeks) for typical lot sizes, amount on Ira nd, and the direct components of each...
-
Explain why, in your opinion, allowing a physician to prescribe lethal medication under aid-in-dying laws is or is not euthanasia.
-
A sinusoidal transverse wave is traveling along a string in the negative direction of an x axis. Figure shows a plot of the displacement as a function of position at time t = 0; the scale of the y...
-
1. What competitive advantage does Hazel have over a professional lawn care service? 2. Hazel would like to increase her profits, but she doesnt believe that it would be wise to raise her prices...
-
Explain why a driving record helps reduce the asymmetric information problems in providing insurance.
-
B, a chemist, was employed by A, a manufacturer, to work on a secret process for As product under an exclusive three-year contract. A employed C, a salesperson, on a week-to-week basis. B and C...
-
Progress On January 1, 2020, Tamarisk SpA had the following equity accounts 1.500.000 Share Capital-Ordinary (25 par value, 60.000 shares issued and outstanding) Share Premium-Ordinary Retained...
-
Explicitly justify the symmetry relations (4.2.4). Note that the first relation follows directly from the symmetry of the stress, while the second condition requires a simple expansion into the form...
-
Verify the curvilinear cylindrical coordinate relations (3.8.5) and (3.8.6). Equation 3.8.5 Equation 3.8.6 V.a , _ 1 + ar r , ar + er T, + + r 1tre 20 1. r 20 + dz -co + atr Zer + z airez+-(a,er +...
-
What is the function of the septum in the heart? What would happen if the heart had no septum?
-
Tabletop Exercise (15%) Develop a tabletop exercise for your organization or community. The size and scope of your exercise can be whatever you need it to be in order for you to complete the...
-
Within the framework of the Porter Five-Forces Model of Competition, describe the competitive force of rivalry among competing sellers. What are some of the factors that increase the rivalry among...
-
The XYZ Corporation has decided to make some changes to help with the work-life balance of its employees. Currently, the organization has 40 employees: 25 full-time employees and 15 part-time...
-
Grocery prices tend to play a role in how people view inflation because of how frequent these purchases are for households. In the past four years grocery prices have jumped 25% which passes overall...
-
Bogg County is a rural area whose residents rely on farming for income. The most popular crop in Bogg County is tobacco, a very labor-intensive plant. To save money, many farmers employ Hispanic...
-
y(t) = t2 sin(t) is a solution of dy/dt = 2t sin(t) + t2 cos(t). Show that the above are solutions of the given differential equation.
-
B made an issue of 150,000 $1 ordinary shares at a premium of 20% the proceeds of which is received by cheque. What is the correct journal to record this? A. Bank Share capital Share premium B. Bank...
-
Heterogeneous equilibria and calcite solubility. If river water in Box 7-2 is saturated with calcite (CaCO 3 ), [Ca 2+ ] is governed by the following equilibria: (a) From these reactions, find the...
-
Using activity coefficients correctly, find the pH of 1.0 10 -2 M NaOH.
-
(Without activities), calculate the pH of (a) 1.0 10 -8 M HBr (b) 1.0 10 -8 M H2SO4 (H2SO4 dissociates completely to 2H + plus SO 2 4 - at this low concentration.)
-
Each week you must submit an annotated bibliography. Entries of current events relating to the economic concepts and the impact on the company or the industry of your company. You must use acceptable...
-
Fluffy Toys Ltd produces stuffed toys and provided you with the following information for the month ended August 2020 Opening WIP Units 5,393 units Units Started and Completed 24,731 units Closing...
-
Part A Equipment 1,035,328 is incorrect Installation 44,672 is incorrect Anything boxed in red is incorrect sents 043/1 Question 9 View Policies Show Attempt History Current Attempt in Progress...

Study smarter with the SolutionInn App


