Explicitly justify the symmetry relations (4.2.4). Note that the first relation follows directly from the symmetry of
Question:
Explicitly justify the symmetry relations (4.2.4). Note that the first relation follows directly from the symmetry of the stress, while the second condition requires a simple expansion into the form ekl = 1/2 (ekl + elk) to arrive at the required conclusion.
Equation 4.2.4
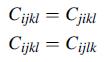
Transcribed Image Text:
Cijkl = Cjikl Cijkl = Cijlk
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
OCunenji C 00 CC C G CC z ex ex C...View the full answer

Answered By

Hemstone Ouma
"Hi there! My name is Hemstone Ouma and I am a computer scientist with a strong background in hands-on experience skills such as programming, sofware development and testing to name just a few. I have a degree in computer science from Dedan Kimathi University of Technology and a Masters degree from the University of Nairobi in Business Education. I have spent the past 6 years working in the field, gaining a wide range of skills and knowledge. In my current role as a programmer, I have had the opportunity to work on a variety of projects and have developed a strong understanding of several programming languages such as python, java, C++, C# and Javascript.
In addition to my professional experience, I also have a passion for teaching and helping others to learn. I have experience as a tutor, both in a formal setting and on a one-on-one basis, and have a proven track record of helping students to succeed. I believe that with the right guidance and support, anyone can learn and excel in computer science.
I am excited to bring my skills and experience to a new opportunity and am always looking for ways to make an impact and grow as a professional. I am confident that my hands-on experience as a computer scientist and tutor make me a strong candidate for any role and I am excited to see where my career will take me next.
5.00+
8+ Reviews
22+ Question Solved
Related Book For 

Elasticity Theory Applications And Numerics
ISBN: 9780128159873
4th Edition
Authors: Martin H. Sadd Ph.D.
Question Posted:
Students also viewed these Engineering questions
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
In Figure 3-4, the current position of the demand curve is D 1 , and the price of a wireless earbud, which is a normal good, is $3. If there is an increase in consumer incomes, will the demand curve...
-
A sinusoidal wave is traveling on a string with speed 40 cm/s. The displacement of the particles of the string at x = 10 cm is found to vary with time according to the equation y = (5.0 cm) sin[1.0 ...
-
Calculate the position of the maximum in the radial distribution function for Li 2+ in its ground state using the wave function in P21.13.
-
Describe a lapping scheme and an audit step to discover lapping.
-
John Palmer and Henry Morrison formed the limited partnership of Palmer & Morrison for the management of the Huntington Hotel and filed an appropriate certificate in compliance with the limited...
-
Please check my answer. I'm not quite sure about my answer. You have just been hired as a loan officer at Westmount Bank. Your supervisor has given you a file containing a request from Hill Company,...
-
Substituting the general isotropic fourth-order form (4.2.6) into (4.2.3), explicitly develop the stressstrain relation (4.2.7). Equation 4.2.6 Equation 4.2.3 Equation 4.2.7 Cik = + ; + jk
-
Show that the components of the C ij matrix in Eq. (4.2.2) are related to the components of C ijkl by the relation: Equation 4.2.2 Cij = C1 C1122 C1133 C1112 C1123 C1131 C2211 C2222 C2233 C2212 C2223...
-
If the Ka for HNO2 is 5.6 104, what is the Kb for NO2 (aq)?
-
What is the present value of $3,000 per year for 9 years discounted back to the present at 11 percent?
-
The next year the common stock of Gold Corp. will pay a dividend of $6.35 per share. If the company is growing at a rate of 5.43 percent per year, and your required rate of return is 13.49 percent,...
-
Minnie wants to buy a home. She contacts the owner of a potential property that she likes. While Minnie is not sure that the home is for her, she wants to prevent someone else from buying the...
-
As employees health changes, they may miss work due to illness or injury increasing healthcare cost for employees and employers. Furthermore, employees may be less productive while at work due to...
-
Camembert's commercial paper is currently selling at a discount. It sells for 98.36 of par and matures in 85 days. a. Calculate its yield as quoted in the market. b. Calculate its effective annual...
-
Clark Heter is an industrial engineer at Lyons Products. He would like to determine whether there are more units produced on the night shift than on the day shift. Assume the population standard...
-
[a] Two foam blocks, each with a charge of 19 micro coulombs (1 C = 10-6 C), are both held in place 19 cm apart in the east-west direction. A foam ball with a charge 49 C is placed 55 cm north of the...
-
Look up the equilibrium constant for the ion-pairing reaction in Appendix J. (a) Use the systematic treatment of equilibrium to find [Zn 2+ ] in 0.010 F ZnSO4. Neglect activity coefficients and any...
-
Use the systematic treatment of equilibrium to find the concentrations of the major species in a saturated aqueous solution of LiF. Consider these reactions: LiF(s) = Li* +F Ksp = [Li*]YLi+[F]YF Kjon...
-
Use the systematic treatment of equilibrium to find the concentrations of the major species in a saturated aqueous solution of LiF. Consider these reactions: (a) Initially, set the ionic strength to...
-
The gross increases in owner's equity attributable to business activities are called Oa. expenses Ob. revenues Oc. liabilities Od. assets 13
-
The gross increases in owner's equity attributable to business activities are called Oa. expenses Ob. revenues Oc. liabilities Od. assets 13
-
Ned's landscaping business has grown so much that he is planning on having a large warehouse built to hold his equipment and supplies. What type of purchase would this represent? Multiple choice...

Study smarter with the SolutionInn App


