A tank with an attached manometer contains water at 20C. The atmospheric pressure is 100 kPa. There
Question:
A tank with an attached manometer contains water at 20°C. The atmospheric pressure is 100 kPa. There is a stopcock located 1 m from the surface of the water in the manometer. The stopcock is closed, trapping the air in the manometer, and water is added to the tank to the level of the stopcock. Find the increase in elevation of the water in the manometer assuming the air in the manometer is compressed isothermally.
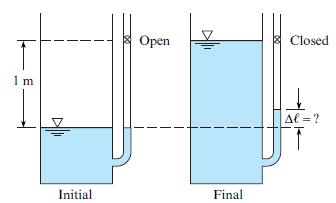
Open Closed 1 m Af =? Initial Final
Step by Step Answer:
The manometer contains air that is compressed isothermally which means that the temperature of the a...View the full answer



Engineering Fluid Mechanics
ISBN: 9781118880685
11th Edition
Authors: Donald F. Elger, Barbara A. LeBret, Clayton T. Crowe, John A. Robertson
Related Video
In this video, the concept of pressure has been explained. The bottle is filled up with water, remove the pipe, and now you can see the clear flow of water from the holes due to liquid pressure.You can see that the second layer is holding the weight of the first layer and the third layer holding the weight of both upper layers, that’s why liquid flows out wider from the third as compared to the first two.At the first hole, water doesn’t roll along the walls; it flows out at an angle.At the second hole, you’ll see that the water flows out at a wider angle as compared to the first hole.At the third hole, we can see that the flow of water is much wider/greater than the other holes.This proves that the pressure at the bottom of the bottle is much more than the pressure at the top layers. P = ????gh
Students also viewed these Engineering questions
-
A top Mt. Everest, the atmospheric pressure is 210 mmHg and the air density is 0.426 kg/m3. (a) Calculate the air temperature, given that the molar mass of air is 29.0 g/mol. (b) Assuming no change...
-
A tank with an internal volume of 1 m3 contains air at 800 kPa and 25C. A valve on the tank is opened allowing air to escape and the pressure inside quickly drops to 150 kPa, at which point the valve...
-
At 20 C the densities of fresh water and ethyl alcohol are, respectively, 998 and 789 kg/m3. Find the ratio of the adiabatic bulk modulus of fresh water to the adiabatic bulk modulus of ethyl alcohol...
-
You have learned a great deal about the Internet Protocol (IP). IP is a set of rules for how data is sent across networks and arrive at the intended destination. An IP address is a numeric identifier...
-
Duke Schmidt has a renters insurance policy for $20,000 of household goods, has selected $300,000 liability with $1,000 deductible, and his credit is rated as occasional payments past 30 days. If...
-
The value of a resistor R is to be determined using the ammetervoltmeter setup shown in Figure P28.63. The ammeter has a resistance of 0.500 Ω, and the voltmeter has a resistance of 20 000...
-
Type in the address www.massdot.state.ma.us/highway/.. TheBigDig.aspx and navigate through the Web site supporting the Boston Tunnel project. Evaluate the performance of this project using the model...
-
Mary Palmquist, a Wall Street securities analyst, wants to determine the relationship between Chile's gross domestic product (GDP) and the profits (after taxes) of the Carlton Company. She obtains...
-
What is the danger in viewing unit costs as variable costs?
-
database management systems in the world. It is a relational database management system (RDBMS) that uses Structured Query Language (SQL) to manage and manipulate data. MySQL was created by Michael...
-
A tank is fitted with a manometer on the side, as shown. The liquid in the bottom of the tank and in the manometer has a specific gravity (SG) of 3.0. Th e depth of this bottom liquid is 20 cm. A 15...
-
Water occupies the bottom 1.2 m of a cylindrical tank. On top of the water is 0.8 m of kerosene, which is open to the atmosphere. If the temperature is 20C, what is the gage pressure at the bottom of...
-
Rank the following carbocations in order of increasing stability: a. b. c.
-
Figure < 4 ft/s 45 0.75 ft 3 ft/s 1.50 ft 1 of 1 < Part A Determine the velocity of point A on the rim of the gear at the instant shown.(Figure 1) Enter the x and y components of the velocity...
-
what ways can leaders facilitate cognitive reframing and emotional regulation techniques to promote constructive conflict resolution ?
-
What is the level of sales needed to achieve a 10% return on an investment of $10,000,000 for a restaurant (the restaurant has main products it sells: food, beverage and gift shop items) and cover...
-
1. An online computer assembling mobile phone Application provides interfaces for end users to assemble computers by selecting computer accessories with different configurations from different...
-
1. (# 3.21, Text) Plot the longitudinal and transverse coefficients of thermal expansion for a unidirectional glass-polyester composite as functions of fiber volume fraction. Assume the following...
-
Xerox Corporation is the company that made the photocopier popular, although it now describes itself as a technology and services enterprise that helps businesses deploy document management...
-
Read Case Study Google: Dont Be Evil Unless and answer the following: Given its mission of providing information to the world, should Google censor searches in China?
-
Use the Transfer Function block to construct a Simulink model to plot the solution of the following equation for 0 t 4. 2x + 12i + 10x Su,(t) Su,(t 2) x(0) = i(0) = 0
-
Use Transfer Function blocks to construct a Simulink model to plot the solution of the following equations for 0 t 2. where f(t) = 75u s (t). 3* + 15x + 18x = f(t) x(0) = i(0) = 0 2 + 16y + 50y =...
-
Use Transfer Function blocks to construct a Simulink model to plot the solution of the following equations for 0 t 2 where f (t) = 50u s (t). At the output of the first block there is a dead zone...
-
Use the following information: \ table [ [ Country , \ table [ [ Consumer Prices ] ] , Interest Rates,Current Units ( per US$ ) ] , [ Forecast , 3 - month, 1 - yx Covt Bond,, ] , [ 2 0 2 4 e ,...
-
Year-to-date, Yum Brands had earned a 3.70 percent return. During the same time period, Raytheon earned 4.58 percent and Coca-Cola earned 0.53 percent. If you have a portfolio made up of 40 percent...
-
Rate of Return If State Occurs State of Probability of Economy State of Economy Stock A Stock B Stock C Boom .15 .31 .41 .21 Good .60 .16 .12 .10 Poor .20 .03 .06 .04 Bust .05 .11 .16 .08 a. Your...

Study smarter with the SolutionInn App


