The equilibrium constant for formation of CuCl 2 - is given by What is the solubility of
Question:
The equilibrium constant for formation of CuCl2- is given by
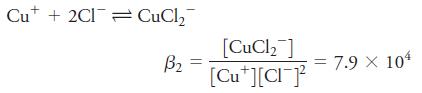
What is the solubility of CuCl in solutions having the following analytical NaCl concentrations:
(a) 5.0 M?
(b) 5.0 3 10-1 M?
(c) 5.0 3 10-2 M?
(d) 5.0 3 10-3 M?
(e) 5.0 3 10-4 M?
Transcribed Image Text:
Cu* + 2Cl= CuCl, [CuCl, ] B2 [Cu*][CI} = 7.9 X 104
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Write equation for complex formation of CuCl 2 where 4 is the complex formation constant for CuCl 2 Cu 3 2Cl CuCl 2 2 CuCl 2 Cu 2 Cl 2 79 10 4 CuCl 2 ...View the full answer

Answered By

Umair Expert
Hi Everyone.
I have 6 years of teaching experience.
I am serving as a tutor 2 more websites.
I will provide you projects and solutions of questions related to any subject.
I am a good programmer.(PHP, Python)
I am good command in mathematics.
I am here to assist you.
I'll be very happy to work with you...
Thanks dear..
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Analytical Chemistry
ISBN: 9780357450390
10th Edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Question Posted:
Students also viewed these Sciences questions
-
The equilibrium constant for the reaction H2 + at 1 atm and 1500C is given to be K. Of the reactions given below, all at 1500C, the reaction that has a different equilibrium constant is (a) H2 + 12O2...
-
The equilibrium constant for the H2 + ½ O2 H2O reaction at 1 atm and 1200 K is KP. Use this information to determine the equilibrium constant for the following reactions: (a) at l atm H, +...
-
The equilibrium constant for the reaction SO3 SO2 + O has the following values: Determine the average heat of dissociation using graphical method. T 800 K 900 K 1000 K 1105K | 0.0319 | 0.153 | 0.540...
-
Hrishi is a senior executive for a large manufacturing company in Mississauga Ontario where he has been employed for the past 10 years and his annual salary is $250,000 including bonus. He is 47...
-
Manuel bought a $100,000 bond with a 4% coupon for $92,300 when it had five years remaining to maturity. What was the prevailing market rate at the time Manuel purchased the bond? (Taken from CIFP...
-
Two sheets of copper of area 1.50 m are separated by 10.0 cm. What is the rate of transfer of heat by conduction from the warm sheet (50e) to the cold sheet (-10C). What is the rate of loss of heat?
-
Wayflaire Inc. produces home products. The cost to manufacture a case ( 36 sets) of its most popular set of casual dinnerware is as follows: The average sales price for a case of dinnerware is \(\$...
-
Every week BosssMustang of Oakland, California, receives shipments from 10 different suppliers in the Los Angeles area. Each suppliers order weighs, on average, 500 pounds. A direct truck shipment...
-
(Related to Checkpoint 12.1) (Calculating project cash flows and NPV) You are considering expanding your product line that currently consists of skateboards to include gas-powered skateboards, and...
-
Write a method called maxToTop that takes a stack of integers as a parameter and moves the largest value in the stack to the top of the stack, leaving all other values in their original order. You...
-
Use activities to calculate the molar solubility of Zn(OH) 2 in (a) 0.0150 M KCl. (b) 0.0250 M K 2 SO 4 . (c) The solution that results when you mix 40.0 mL of 0.250 M KOH with 60.0 mL of 0.0250 M...
-
For a solution in which = 6.5 x 10 -2 , calculate K' sp for (a) AgSCN. (b) PbI 2 . (c) La(IO 3 ) 3 . (d) MgNH 4 PO 4 .
-
Use the algebraic tests to check for symmetry with respect to both axes and the origin. 1. x2 y = 0 2. x y2 = 0 3. y = x3
-
Nitrogen (N) contained in a piston-cylinder arrangement, initially at 4.9 bar and 425 K, undergoes an expansion to a final temperature of 300 K, during which the pressure-volume relationship is pv1.3...
-
You are required to write an interactive C++ program that prompts the user for two integers X and Y and performs the following tasks: Adds two numbers. Subtracts Y from X. Multiplies X and Y. Divides...
-
b. It is suspected that a small number of components in a large batch are faulty. The proportion of faulty components is estimated to be 1 in 500. A sample of 100 components are extracted. i. Justify...
-
The indifference utility function is given as U(E(r), o) = E(r) - 0.5Ao, where A is a risk aversion coefficient (i) How do we determine the optimal portfolio consisting of two stocks that maximizes...
-
Suppose an inverse demand function is given by p = 1 0 qd and an inverse supply function is given by p = 4 + 2 qs . ( a ) What is the equilibrium price and quantity combination? [ 5 ] ( b )...
-
On June 30, 2018, Plaster, Inc., paid $916,000 for 80 percent of Stucco Company's outstanding stock. Plaster assessed the acquisition-date fair value of the 20 percent noncontrolling interest at...
-
Uniform electric field in Figure a uniform electric field is directed out of the page within a circular region of radius R = 3.00 cm. The magnitude of the electric field is given by E = (4.50 x 10-3...
-
Calculate the theoretical cell potential of the following cells. If the cell is short-circuited, indicate the direction of the spontaneous cell reaction. (a) Zn ( Zn2+ (0.1000 M) (( Co2+ (5.87 ( 10-4...
-
Calculate the potential of the following two half-cells that are connected by a salt bridge: (a) A galvanic cell consisting of a lead electrode (right electrode) immersed in 0.0220 M Pb2+ and a zinc...
-
Write balanced net ionic equations to describe (a) The oxidation of Mn2+ to MnO4- by ammonium peroxydisulfate. (b) The oxidation of U4+ to UO22+ by H2O2. (c) The titration of H2O2 with KMnO4.
-
ACCT 597 Assignment 5 The KJA partnership is formed by Kennedy, Jane, and Allyson. Kennedy will contribute $500,000 cash as will Jane. Allyson will contribute real property with a FMV of $800,000 and...
-
35. Devin and Merri are getting prequalified. Devin earns $1.300 a month and Merri earns $1,000 a month. Merri receives child support of $700 a month from an ex-husband. Devin gets $150 annual...
-
I need the answer and the work Fox's Pizza currently pays no dividends on its stock. Management decides to issue dividends of $2.15 per share three years from now and expect to increase dividends by...

Study smarter with the SolutionInn App


