A gas has C P * = 35 J/mol K, and follows the equation of state:
Question:
A gas has CP* = 35 J/mol · K, and follows the equation of state: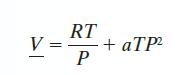 with a = 0.15 cm3/mol · bar2 · K. Find the change in A when the gas is compressed isothermally from T = 300 K and P = 1 bar to P = 10 bar.
with a = 0.15 cm3/mol · bar2 · K. Find the change in A when the gas is compressed isothermally from T = 300 K and P = 1 bar to P = 10 bar.
Transcribed Image Text:
V= RT P + aTP²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
The change in Helmholtz free energy AA for an isotherm...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
An economy is initially described by the following equations C = 500 +0.50(Y-T) 1 = 500-25r d =Y - 25r G = 100 T = 500 M = 2000 P = 4 a. Write down the equations for the IS and LM curves and...
-
A gas has an ideal gas heat capacity of C P * = (7/2)R and is described by the equation of state: Z = 1 + (CP 2 )/(RT) with C = 100 cm 3 /bar mol. A . Find a general expression for the residual...
-
A gas with a flow rate of 300 mol/min enters a steady-state, adiabatic nozzle with negligible velocity at T = 500 K and P = 10 bar and leaves the nozzle at P = 1 bar. The gas has C P * = 40 J/mol K....
-
Name: 9. 11. Bell: Directions: Evaluate each expression using the order of operations. 1. 17-5-4+2 2. 40-32+8+5--2 Date: 5. 2018-(5+3)+7] 3.35-14+2+8 7.1+(-2-5)+(14-17)-4 25 +8+3 4+3 Unit 1: Algebra...
-
Assume you are a member of an international policy setting committee and are responsible for harmonizing audit report requirements internationally. Examine Exhibit 9-8. Based on the varying...
-
Refer to Exercise 8 (hedge fund returns and administrator compensation). The estimated regression equation turned out to be y = 2.2 + .55x. Show the 95% prediction interval estimate of the...
-
For a sport of your choice, construct a plan to indicate where you should gather information from in order to that the management of this sport remains up to date and relevant.
-
Sharp Outfits is trying to decide whether to ship some customer orders now via UPS or wait until after the threat of another UPS strike is over. If Sharp Outfits decides to ship the requested...
-
please solve it fast ASAP One of the indirect costs of bankruptcy is the incentive toward underinvestment, which generally would result in O a. The firm turning down positive NPV projects that would...
-
Imagine a compound has T C = 500 K and P C = 20 bar. Use the Peng-Robinson equation throughout this problem. A. Plot P- V at T = 400 K, T = 500 K, and T = 600 K, assuming the compound has = 0. B....
-
Steam is heated from an initial condition of saturated steam at P = 1.5 bar to a final state of P = 3 bar and T = 3008C. Use the steam tables to find the change in for this process.
-
List and explain some fundamental issues of behavioral finance.
-
Question 3 (15 marks) Sporty Ltd. produces scooters and skateboards. At the beginning of the year, the following volume of activities were budgeted for the year: Production volume/units Direct labour...
-
Consider a rigid body B with center of mass point B*. A set of coordinate axes is chosen centered at B* and defined by mutually orthogonal unit vectors 61, 62, 63 which are fixed in B. The rigid body...
-
Let A be the matrix 1 0 2 4 1 -6 = 7-4 7 -5 3 (a) (2 points) What must a and b be in order to define the linear transformation T: RR by T(x) = Ax. (b) (3 points) What is the image of the vector 2] 1...
-
write a title Understanding the roots of modern educational practices can provide valuable insights into their effectiveness and potential for improvement. One such root influencing contemporary...
-
State the limit for each of the following using the graph. -6. -5 + -3- 3 -2 -2 0 2 -2- w. 3 4
-
What keeps a cloud from falling? A cumulus (fair-weather) cloud consists of tiny water droplets of average radius 5.0 m. Find the terminal velocity for these droplets at 20 C, assuming viscous drag....
-
Determine two different Hamilton circuits in each of the following graphs. A B F G
-
Use MATLAB to solve the following problem: x - 3y = 2 x + 5y = 18 4x - 6y = 10
-
a. Use MATLAB to find the coefficients of the quadratic polynomial y = ax 2 + bx + c that passes through the three points (x, y) = (1, 4), (4, 73), (5, 120). b. Use MATLAB to find the coefficients of...
-
Use the MATLAB program given in Table 8.52 to solve the following problems: a. Problem 3d d. b. Problem 11 Solve the following equations: c. Problem 14 Use MATLAB to solve the following problem: x -...
-
Sunland Company uses the percentage of sales method for recording bad debts expense. For the year, cash sales are $611000 and credit sales are $2790000. Management estimates that 5% is the sales...
-
Michael will earn $148,000 in 2020. a. How much in total will be paid into the entire OASDI system (including Medicare) on behalf of this income? ___________________________ b. How much will Michael...
-
Cost = 125 000 Resdiual Value = 5000 Useful life = 10 years .. using both stright line and double declining method determine 1. Accumlated depreciation at the end of year 2 2. Book value at the...

Study smarter with the SolutionInn App


