A liquid enters an adiabatic throttling valve, illustrated in Figure 6-4, at T = 80F and P
Question:
A liquid enters an adiabatic throttling valve, illustrated in Figure 6-4, at T = 80°F and 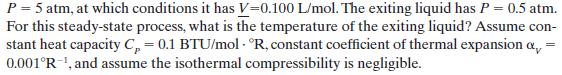
Transcribed Image Text:
P = 5 atm, at which conditions it has V-0.100 L/mol. The exiting liquid has P = 0.5 atm. For this steady-state process, what is the temperature of the exiting liquid? Assume con- stant heat capacity C = 0.1 BTU/mol R, constant coefficient of thermal expansion a 0.001R-1, and assume the isothermal compressibility is negligible. =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
Step 1 Define a system and simplify the energy balance This is another JouleThomson expansion carrie...View the full answer

Answered By

Arshad Ahmad
Well, I am really new to tutoring but I truly believe a good student can be a better teacher. I have always been a topper at school. I passed my Chartered Accountancy at a very young age of 23, a rare feat for most of the students. I am really dedicated to whatever work I do and I am very strict regarding deadlines. i am always committed and dedicated to whatever work allotted to me and I make sure it is completed well within deadline and also I try to give my best in whatever I do. Hope we will have a good time studying together.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Steam is produced at 30 bar and some unknown temperature. A small amount of steam is bled off and goes through an adiabatic throttling valve to 1 bar. The temperature of the steam exiting the...
-
Steam is produced at 70 bar and some unknown temperature. A small amount of steam is bled off just before entering a turbine and goes through an adiabatic throttling valve to atmospheric pressure....
-
Determine the void ratio of a soil if its bulk density is 2.06 g/cc and it has a moisture content of 24 percent. The specific gravity of the soil particles is 2.75. Also, determine its dry density...
-
Gross domestic product (GDP) is a major indicator of a nation's overall economic activity. It consists of personal consumption expenditures, gross domestic investment, net exports of goods and...
-
Use the template for the ABC Corp. valuation in section 2.7 to value Cisco stock. Assume that the weighted average cost of capital for Cisco is 12.6%, the growth rate for years 15 is 4%, and that the...
-
Adjusting entries are often required in accounting. Please describe the various types of adjusting entries and give at least three examples.
-
Randolph College and Salem College are within 20 miles of each other, and the students at each college frequently date. The students at Randolph College are debating how good their dates are at Salem...
-
IBM Corporation was the leading manufacturer of electronic computers in the world. Based on its internal memoranda, IBM's long-run total cost of producing various quantities of its Pisces (370/168)...
-
100 kg/min of liquid nitrogen is produced by the steady-state process shown in Figure 5-17. 1. Nitrogen enters the process at P = 1 bar and T = 300 K. 2. The nitrogen is cooled in a heat exchanger...
-
Describe how liabilities are reported and analyzed.
-
In the two-mesh network shown in Fig. 16-15 there is no initial charge on the capacitor. Find the loop currents i 1 and i 2 which result when the switch is closed at t = 0. 50 v + 10 i Fig. 16-15 i...
-
Colson Inc. declared a $ 3 2 0 , 0 0 0 cash dividend. It currently has 1 2 , 0 0 0 shares of 7 % , $ 1 0 0 par value cumulative preferred stock outstanding. It is two years in arrears on its...
-
Discuss the origins of the American legal system, including the common law, precedent, stare decisis, and judicial review. Please provide as much details as possible.?
-
Discuss naive empiricism? *Also, do you think this method is effective to reach any logical conclusion in order to examine common law theories or principles.
-
Describe the role of both the judge and the jury in criminal trials. Explain why the jury is both substantively and symbolically important. Please provide as much details as possible.?
-
What are the principal advantages of utilizing functions in software engineering, and how do they contribute to code modularity and maintainability?
-
In nuclear fission, a nucleus of uranium-238, which contains 92 protons, can divide into two smaller spheres, each having 46 protons and a radius of 5.90 & 10'15 m. What is the magnitude of the...
-
Nitrogen monoxide reacts with hydrogen as follows: 2NO(g)+ H2(g) N2O(g) + H2O(g) The rate law is [H2]/ t = k[NO]2[H2], where k is 1.10 107 L2/(mol2s) at 826oC. A vessel contains NO and H2 at...
-
What will be the value of the destination operand after each of the following instructions execute? .data var1 BYTE var2 WORD var3 SWORD var4 DWORD .code 7, 6, 0Fh, 3 2122h, 9396h, OF10Dh, 9527h -55,...
-
Paul corporation of profit, maximizing monopoly. It sells a patented rabies vaccine for pets and earns economic profits.
-
Why does it later rise? How would you describe that in economic terms again?

Study smarter with the SolutionInn App


