Find the molar volume of nitrogen at P = 10 bar and T = 330 K, using
Question:
Find the molar volume of nitrogen at P = 10 bar and T = 330 K, using the following methods.
A. The Soave equation of state
B. The Peng-Robinson equation of state
C. The virial equation of state
D. The Lee-Kesler generalized correlation
E. Appendix F
Appendix F
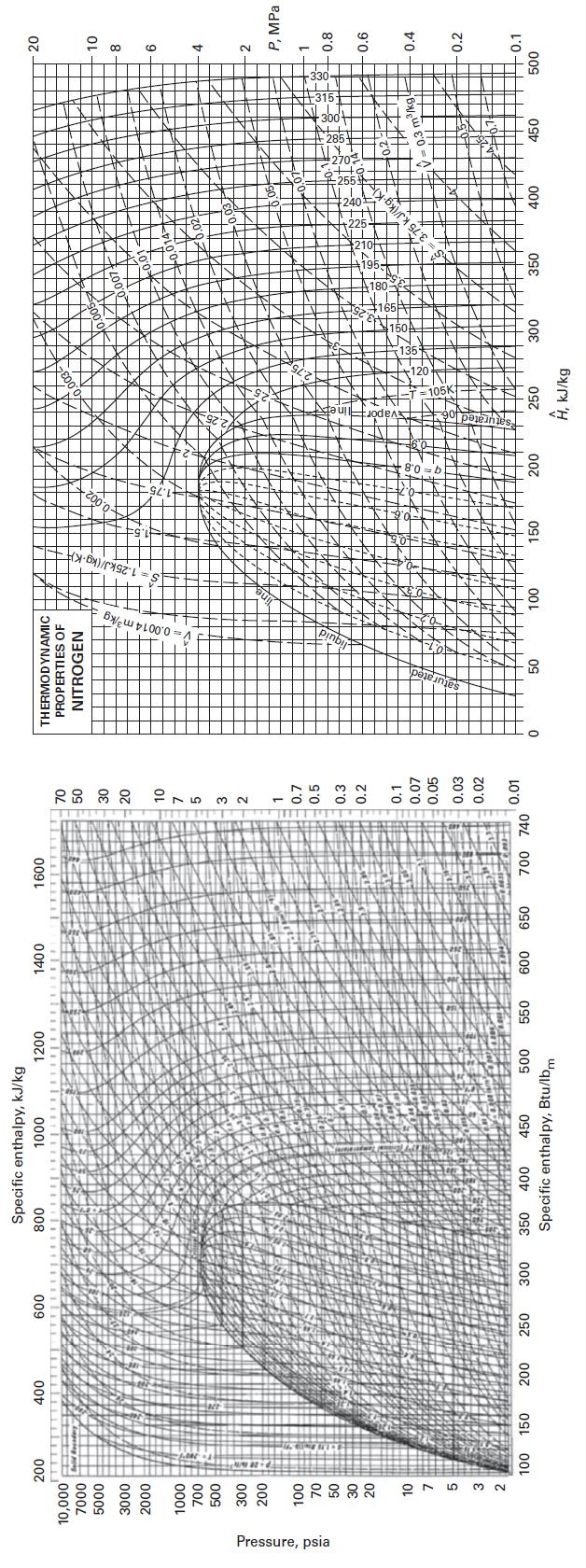

Transcribed Image Text:
Pressure, psia 200 10,000 7000 5000 3000 2000 1000 700 500 300 200 100 70 50 30 20 10 3 2 100 150 400 200 600 250 300 Specific enthalpy, kJ/kg 1000 800 1200 350 400 450 500 Specific enthalpy, Btu/lbm 550 1400 600 650 1600 700 740 70 50 30 20 10 7 5 3 2 1 0.7 0.5 0.3 0.2 0.1 0.07 0.05 0.03 0.02 0.01 0 THERMODYNAMIC PROPERTIES OF NITROGEN 50 <> 100 150 200 250 Ĥ, kJ/kg 300 350 400 0.3 m³/kg 450 20 10 8 6 4 2 P, MPa 1 0.8 0.6 0.4 0.2 0.1 500
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)

Answered By

Bhartendu Goyal
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions. I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and help them achieve great subject knowledge. I have expertise in computer subjects like C++, C, Java, and Python programming and other computer Science related fields. Many of my student's parents message me that your lessons improved their children's grades and this is the best only thing you want as a tea...
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
Find the molar volume of Freon 22 at P = 5 bar and T = 20C (it is a vapor at these conditions), using the following methods. A. The Soave equation of state B. The Peng-Robinson equation of state C....
-
Find the molar volume of methane at P = 15 bar and T = 200C (it is a gas at these conditions), using the following methods. A. The Soave equation of state B. The Peng-Robinson equation of state C....
-
Question 3. Find the second derivative of y = x - 1 Question 4. If f(x) = x-1 Find f'(-2).
-
Which one of the following results from the latest decision round are least important in providing guidance to company managers in making their strategic moves and decisions to improve their...
-
Foreign exchange rates are used to establish budgets and track actual performance. Of the various exchange rate combinations mentioned in this chapter, which do you favor? Why? Is your view the same...
-
In Problem 13.7 on page 470, you used the total number of staff present and remote hours to predict standby hours (stored in Standby). Using the results from that problem, a. Construct a 95%...
-
17.7 Barfield Primrose Limited is the manufacturer of the renowned 'Primrose' ice cream maker, which retails at 199. Barfield Primrose sells to wholesalers at 145 per unit. The prime cost structure...
-
Fill in the missing particles or nuclei; (a) n + 137/56Ba ? + y; (b) n + 137/56Ba 137/55Cs + ?; (c) d + 2/1H 4/2 He + ?; (d) + 197/79Au ? + d where d stands for deuterium.
-
A patio set cost a retailer $419.00 less 25 3/4%, 18%, 7%. Markup is of the regular selling price. During the end-of-season sale, the is marked down (a) What is the end-of-season sale price? (b) What...
-
Use the Joback method to estimate T c and P c of each of the following compounds. A. Butane B. 1-Hexanol C. 2-chloropentane D. 3-Hexene E. 1,3-butadiene
-
Using data in Appendix C-1, determine the Peng-Robinson parameters a and b for each of the following compounds at the temperature T = 100C. A. Ethane B. Acetone C. Benzene D. Toluene E. Decane Name...
-
Sheena Stone 1S an attorney in Los Angeles. Stone uses the direct write-off method to account for uncollectible receivables. At November 30, 2010, Stones accounts receivable totaled \($21,000\)....
-
McDonald's conducts operations worldwide and is managed in two primary geographic segments: US, and International Operated Markets, which is comprised of Australia, Canada, France, Germany, Italy,...
-
Woodland Wearables produces two models of smartwatches, the Basic and the Flash. The watches have the following characteristics:Basic Flash Selling price per watch$ is 270$ 460 Variable cost per...
-
Based on the information provided and recognizing the value of coordinating across its portfolio of businesses, how should LendingTree manage these newer businesses? * as more integrated units * as...
-
Trust Fund Worksheet Background An inter vivos trust was created by Isaac Posney. Isaac owned a large department store in Juggins, Utah. Adjacent to the store, Isaac also owned a tract of land that...
-
A popular theory is that presidential candidates have an advantage if they are taller than their main opponents. Listed are heights (in centimeters) of randomly selected presidents along with the...
-
A nurse applies a force of 4.40 N to the piston of a syringe. The piston has an area of 5.00 105 m2. What is the pressure increase in the fluid within the syringe?
-
State whether each statement is true or false. If false, give a reason. {purple, green, yellow} = {green, pink, yellow}
-
Suppose that data show that a certain stock price is normally distributed with a mean of $150 and a variance of 100. Create a simulation to compare the results of the following two strategies over...
-
Write a script le to simulate 100 plays of a game in which you ip two coins. You win the game if you get two heads, lose if you get two tails, and ip again if you get one head and one tail. Create...
-
Write a script le to play a simple number guessing game as follows. The script should generate a random integer in the range 1, 2, 3, . . . , 14, 15. It should provide for the player to make repeated...
-
What is Toms total amount of gross income if: 1) he earned $100 in wages; 2) his credit card forgave his credit card balance of $200; 3) he received a $300 cash gift from his aunt; and 4) and...
-
Carter Co. (a C Corp.), who is a calendar year taxpayer received a check for $180,000 in payment for services rendered as a commissioned sales agent near the end of 2017. In 2018, the customer...
-
spot 5 mistakes in this insurance document Second, spot 5 mistakes in the life insurance policy attached to this document, Insurance Company: 222 nurance Client: Mike Swanson Address 1 231 Ocean St....

Study smarter with the SolutionInn App


