For an ethylene glycol n-butyl ether (1) + water (2) system at 310 K with 70% by
Question:
For an ethylene glycol n-butyl ether (1) + water (2) system at 310 K with 70% by mass water, determine if the system is one stable liquid phase or two stable liquid phases at equilibrium. If the latter, provide the mass fraction of the co-existing phases.
figure 13-3
Transcribed Image Text:
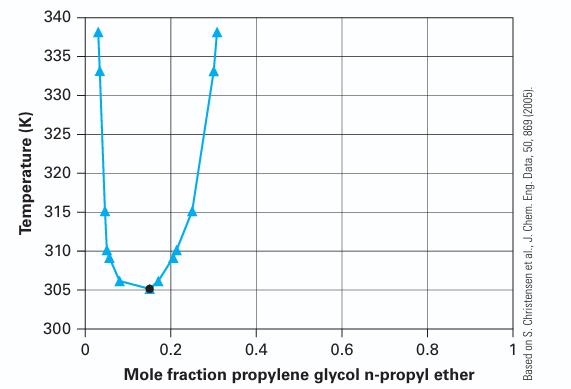
Temperature (K) 340 335 330 325 320 315 310 305 300 0 0.2 0.4 0.6 0.8 Mole fraction propylene glycol n-propyl ether Based on S. Christensen et al., J. Chem. Eng. Data, 50, 869 (2005).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
Figure 1.28 shows a circuit with five elements. If P1 = 205 W, P2 = 60W, P4 = 45W, P5 = 30W, calculate the power p3 received or delivered by element 3. 3
-
Select all that is /are true about the financial markets. A. Financial markets bring together the buyers and sellers of debt and equity together . B. Stocks trading on an organized exchange such as...
-
Amortization of Bond Premium or Discount The appropriate method of amortizing a premium or discount on issuance of bonds is the effective interest method. Required: 1. What is the effective interest...
-
Use the Ratio Test to determine if each series converges absolutely or diverges. 8 n=1 nt (-4)"
-
Why is feedback a necessary component of the communication process?
-
Without using Fig. 13.3, predict which bond in each of the following groups is the most polar. a. COF, SiOF, GeOF b. POCl, SOCl c. SOF, SOCl, SOBr d. TiOCl, SiOCl, GeOCl e. COH, SiOH, SnOH f. AlOBr,...
-
Use the general solution in Perry's Chemical Engineers' Handbook (Wankat and Knaebel, 2019, p. 5-47) to solve the problem of a dissolving solid in a concentrated fluid using the known ratio of...
-
The statement of cash flows for Riker Company (prepared using the indirect method) follows. Consider the following additional information: (a) Sales for the year totaled $812,350. Cost of goods sold...
-
K Calculate the monthly payments of a 30-year fixed-rate mortgage at 6.75 percent for $149,700 How much interest is paid over the life of the loan? Note: Round intermediate computations to at least...
-
This problem expands upon Example 15-4. A reaction vessel is rigid and has a volume of 500 L and initially contains 10 moles of o-xylene. The liquid phase is exposed to catalyst that facilitates...
-
30 mol/s of hydrogen gas and 15 mol/s of air, each compressed to 25 bar, enter a steady state reactor as shown in Figure 15-5, where the nitrogen in the air reacts with the hydrogen to form ammonia:...
-
Serges is the owner of a retail meat marketing business. Without authority his managing agent borrowed $3,500 from David on Sergess behalf, for use in Sergess business. Serges paid $200 on the...
-
A continuous system has an infinite number of natural frequencies. Indicate whether the statement presented is true or false. If true, state why. If false, rewrite the statement to make it true.
-
What is the signature of the Fourier transform? A DOF system with a cubic nonlinearity has a linear natural frequency of \(120 \mathrm{rad} / \mathrm{s}\). The system is forced by harmonic...
-
A continuous system is also referred to as a distributed parameter system. Indicate whether the statement presented is true or false. If true, state why. If false, rewrite the statement to make it...
-
Explain why a superharmonic resonance occurs. A DOF system with a cubic nonlinearity has a linear natural frequency of \(120 \mathrm{rad} / \mathrm{s}\). The system is forced by harmonic excitations...
-
Mode shapes corresponding to distinct natural frequencies of a continuous system are orthogonal with respect to the potential-energy scalar product. Indicate whether the statement presented is true...
-
The probability that the loss from a portfolio will be greater than $10 million in one month is estimated to be 5%. (a) What is the one-month 99% VaR assuming the change in value of the portfolio is...
-
The registrar of a college with a population of N = 4,000 full-time students is asked by the president to conduct a survey to measure satisfaction with the quality of life on campus. The following...
-
The following data are available for a gas: To calculate the molar volume at 25 C, 12 bar, our team came up with two different suggestions: (a) Linear interpolation for Vbetween the given pressure, ...
-
Determine the temperature and phase of water from the following information. If the phase is a vapor-liquid mixture, report the fraction of vapor and liquid. a) The specific volume of water is 100 cm...
-
In 1656, Otto von Guericke of Magdeburg presented his invention, a vacuum pump, through a demonstration that became a popular sensation. A metal sphere made of two hemispheres (now known as the...
-
Caspian Sea Drinks is considering the purchase of a new water filtration system produced by Rube Goldberg Machines. This new equipment, the RGM - 7 0 0 0 , will allow Caspian Sea Drinks to expand...
-
RCD sells cars in R-land, where the currency is the R$. At the start of 20X2, RTW introduced more favourable credit terms and, as a result, credit sales have increased from R$800,000 in 20X1 to...
-
1.Does the organization have qualified employees to assess business income tax? 2.Do the annual plan prepared by the sub city match with the actual performance? 3.Do tax payers maintain book of...

Study smarter with the SolutionInn App


