Use a Lagrange multiplier technique to show that for a system characterized by the following modified Maxwell-Boltzmann
Question:
Use a Lagrange multiplier technique to show that for a system characterized by the following modified Maxwell-Boltzmann distribution function
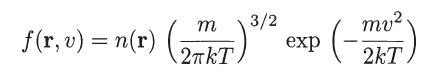
where Tis constant, the entropy S, defined by
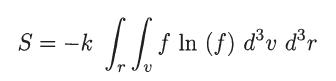
is a maximum when the density n is constant, independent of r. Consider that the system has a total of N particles in a fixed volume V at a temperature T.
Transcribed Image Text:
m f(x,+) = n(x) (2%r)*/2 = exp mv². 2kT
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
To show that the entropy S is maximized when the density n is constant and independent of r we need ...View the full answer

Answered By

Rinki Devi
Professional, Experienced, and Expert tutor who will provide speedy and to-the-point solutions.
Hi there! Are you looking for a committed, reliable, and enthusiastic tutor? Well, teaching and learning are more of a second nature to me, having been raised by parents who are both teachers. I have done plenty of studying and lots of learning on many exciting and challenging topics. All these experiences have influenced my decision to take on the teaching role in various capacities. As a tutor, I am looking forward to getting to understand your needs and helping you achieve your academic goals. I'm highly flexible and contactable. I am available to work on short notice since I only prefer to work with very small and select groups of students.
I have been teaching students for 5 years now in different subjects and it's truly been one of the most rewarding experiences of my life. I have also done one-to-one tutoring with 100+ students and helped them achieve great subject knowledge.
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The text derives a decomposition of a particular type of chooser option into a call maturing at time T 2 and a put maturing at time T 1 . By using putcall parity to obtain an expression for instead...
-
Use the recursion formula of Exercise 5.8 to show that for θ = 12 the binomial distribution has (a) A maximum at x = n/2 when n is even; (b) Maxima at x = n 1 / 2 and x = n + 1 / 2 when...
-
In Corollary 10.2 we were concerned with finding the appropriate "big-Oh" form for a function f: Z+ R+ U {0} where f(1) ¤ c, for c Z+ f(n) ¤ af (n / b) + c, for a, b Z+ with b ¥ 2,...
-
Segment Analysis Winston Sylvester Corporation is a brokerage and financial service company. The company recently provided information about its major business segments as follows (in millions):...
-
Preferred Products has issued preferred stock with an $8 annual dividend that will be paid in perpetuity. a. If the discount rate is 12%, at what price should the preferred sell? b. At what price...
-
Which is the best example of a supervisor making feedback descriptive rather than evaluative? (a) You are a slacker. (b) You are not responsible. (c) You cause me lots of problems. (d) You have been...
-
The General Social Survey. The General Social Survey places much emphasis on asking many of the same questions year after year. Why do you think it does this?
-
Best Corporation acquired 100 percent of the voting common stock of Flair Company on January 1, 20X7, by issuing bonds with a par value and fair value of $670,000 and making a cash payment of...
-
1A) Matt Clark, an auditor with Grant CPAs, is performing a review of Parson Company's inventory account. Parson did not have a good year and top management is under pressure to boost reported...
-
Consider a plasma in which the electrons and the ions are characterized, respectively, by the following distribution functions: (a) Calculate the difference (b) Show that this plasma of electrons and...
-
Consider the case of Maxwell molecules, for which the interparticle force is of the form where K is a constant. (a) Without specifying the form of the distribution functions f (v) and f (v 1 ) for...
-
Find the standard equation of the sphere with the given characteristics. Endpoints of a diameter: (-2, 4, -5), (-4, 0, 3)
-
Identify at least two business systems that support the development of effective work relationships Briefly explain how each system supports the development of effective work relationships.
-
Power and Influence Personal Plan - How will you navigate the realms of power and influence? Why is this personal plan important for you? What do you want to achieve? do a table with SMART goals -...
-
A single-stage trickling-filter plant is proposed for treating a dilute wastewater with a BOD concentration of 170 mg/L. The plant is located in a warm climate, and the minimum wastewater temperature...
-
For the first assignment for this course, compose a written document that contains the following: A description and assessment of your past experiences with policy and program planning, either your...
-
What are the key motivators driving consumer purchasing decisions in our industry? How do consumers perceive our brand compared to competitors, and what factors influence brand loyalty?
-
The following transactions of Plymouth Pharmacies occurred during 2017 and 2018: 2017 Jan. 9 Purchased computer equipment at a cost of $12,000, signing a six-month, 9% note payable for that amount....
-
Quadrilateral EFGH is a kite. Find mG. E H <105 G 50 F
-
For each of the following descriptions draw the structure of a compound that fits the description. (Note: There are many correct answers for each of these problems.) a) An alkyl halide that produces...
-
Assuming that H f is constant in the interval 275 K 600. K, calculate G for the process *H2O, g, 298 K) (H 2 O, g, 600, K). Calculate the relative change in the Gibbs energy.
-
How many different alkenes will be produced when each of the following substrates is treated with a strong base? a) 1-Chloropentane b) 2-Chloropentane c) 3-Chloropentane d) 2-Chloro-2-methylpentane...
-
1,600 Balance Sheet The following is a list (in random order) of KIP International Products Company's December 31, 2019, balance sheet accounts: Additional Paid-In Capital on Preferred Stock $2,000...
-
Question 3 4 pts 9 x + 3 x 9 if x 0 Find a) lim f(x), b) lim, f(x), C), lim , f(x) if they exist. 3 Edit View Insert Format Tools Table : 12pt M Paragraph B IV A2 Tv
-
Mr. Geoffrey Guo had a variety of transactions during the 2019 year. Determine the total taxable capital gains included in Mr. Guo's division B income. The transactions included: 1. On January 1,...

Study smarter with the SolutionInn App


