Numerical evaluation of the effective densities of states of Ge, Si, and GaAs. Calculate the effective densities
Question:
Numerical evaluation of the effective densities of states of Ge, Si, and GaAs. Calculate the effective densities of states in the conduction and valence bands of germanium, silicon and gallium arsenide at 300 K. Note in analogy to Eq. (5.55) we have
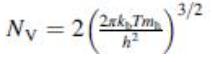
Transcribed Image Text:
Ny = 2( 3/2 2xk Tm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
The effective densities of states in the conduction and valence bands of germanium Ge silicon Si and ...View the full answer

Answered By

Dulal Roy
As a tutor, I have gained extensive hands-on experience working with students one-on-one and in small group settings. I have developed the ability to effectively assess my students' strengths and weaknesses, and to customize my teaching approach to meet their individual needs.
I am proficient at breaking down complex concepts into simpler, more digestible pieces, and at using a variety of teaching methods (such as visual aids, examples, and interactive exercises) to engage my students and help them understand and retain the material.
I have also gained a lot of experience in providing feedback and guidance to my students, helping them to develop their problem-solving skills and to become more independent learners. Overall, my hands-on experience as a tutor has given me a deep understanding of how to effectively support and encourage students in their learning journey.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
The energy gap between valence and conduction bands in germanium is 0.72eV. What range of wavelengths can a photon have to excite an electron from the top of the valence band into the conduction hand?
-
The gap between valence and conduction bands in silicon is 1.12eV. A nickel nucleus in an excited state emits a gamma-ray photon with wavelength 9.31 x 10-4nm. How many electrons can be excited from...
-
The gap between valence and conduction bands in diamond is 5.47eV. (a) What is the maximum wavelength of a photon that can excite an electron from the top of the valence band into the conduction...
-
The following are selected 2023 transactions of Ayayai Corporation. Sept. 1 Oct. 1 1 Purchased inventory from Indigo Ltd. on account for $47,200. Ayayai uses a periodic inventory system. Issued a...
-
Fellowes and Associates Chartered Accountants is a successful mid-tier accounting firm with a large range of clients across Canada. During 2011, Fellows and Associates gained a new client, Health...
-
What are certification and accreditation when applied to information systems security management? List and describe at least two certification or accreditation processes.
-
What is the strong point of the APT compared with the CAPM? And the weak point?
-
In 2018, Bratten Fitness Company made the following cash purchases: 1. The exclusive right to manufacture and sell the X-Core workout machine from Symmetry Corporation for $200,000. Symmetry created...
-
Part 2 of 5 (b) Compute the value of the test statistic. Round the answer to at least two decimal places. z=-2.16 Part: 2/5 Part 3 of 5 (c) Compute the P-value. Round the answer to at least four...
-
The following facts pertain to Lifecycle Corporation: ? Able owns a parcel of land (land A) having a $30,000 FMV and $16,000 adjusted basis. Baker owns an adjacent parcel of land (Land B) having a...
-
Plot of the Fermi distribution function at two different temperatures. Calculate the Fermi function at 6.5 eV if E F = 6.25 eV and T = 300 K. Repeat for T =950 K assuming that the Fermi energy does...
-
Density of states of a piece of Si. Calculate the number of states per unit energy in a 100 by 100 by 10 nm piece of silicon (m* = 1.08 m 0 ) 100 meV above the conduction band edge. Write the results...
-
Would you prefer to hire employees who are high in learning goal orientation or performance goal orientation? Why?
-
Betram Chemicals Company processes a number of chemical compounds used in producing industrial cleaning products. One compound is decomposed into two chemicals: anderine and dofinol. The cost of...
-
1.We have 4 ways to recruit operators for out factory: on line internet, head hunters, ads in trade journals, and on-line company intranet.HR has discovered on-line internet ads reach 300 people per...
-
If Current liabilities Rs . 4 0 , 0 0 0 ; Current assets Rs . 1 , 0 0 , 0 0 0 ; Inventory Rs . 2 0 , 0 0 0 . Find the Quick ratio ? ?
-
Herman Melville\'s Bartleby , the Scrivener is as much about the narrator as it is about the title character himself. As he attempts to learn more and more about Bartleby, he discovers that Bartleby...
-
The New York City Council is ready to vote on two bills that authorize the construction of new roads in Manhattan and Brooklyn. If the two boroughs join forces, they can pass both bills, but neither...
-
Audit standards indicate that there is a presumption that auditors will confirm accounts receivable unless the balance is immaterial, confirmations are deemed ineffective, or the auditors' assessment...
-
In each of the following independent cases, document the system using whatever technique(s) your instructor specifies. a. Dreambox Creations (www.dreamboxcreations.com/) in Diamond Bar, California,...
-
Calculate the pH of a solution made by mixing 50.00 mL of 0.100 M NaCN with (a) 4.20 mL of 0.438 M HClO 4 (b) 11.82 mL of 0.438 M HClO 4 (c) What is the pH at the equivalence point with 0.438 M HClO...
-
Calculate the pH when 25.0 mL of 0.020 0 M 2-aminophenol have been titrated with 10.9 mL of 0.015 0 M HClO 4 .
-
Find the equilibrium constant for the reaction of MES (Table 8-2) with NaOH.
-
Compute the missing amount in the accounting equation for each entity from the financial information? presented: AssetsLiabilitiesEquityFresh Produce$ ?$40,000$26,000Hudson Gift and...
-
XYZ Insurance Company maintains an extensive computer database with information about its policyholders. Data for new policyholders is constantly added and data for existing policyholders is updated....
-
An advantage of using a cold site as a recovery option is less expensive recovery option. Question 9 options: True False

Study smarter with the SolutionInn App


