A mass-loaded piston/cylinder shown in Fig. P4.154, containing air, is at 300 kPa, 17C with a volume
Question:
A mass-loaded piston/cylinder shown in Fig. P4.154, containing air, is at 300 kPa, 17◦C with a volume of 0.25 m3, while at the stops V = 1 m3. An air line, 500 kPa, 600 K, is connected by a valve that is then opened until a final inside pressure of 400 kPa is reached, at which point T = 350 K. Find the air mass that enters, the work, and the heat transfer.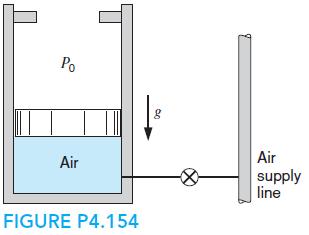
Transcribed Image Text:
Po Air Air supply line FIGURE P4.154
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (6 reviews)
The air mass that enters the pistoncylinder can be found using the Ideal Gas Law PV nRT At the initi...View the full answer

Answered By

Akshay Shete
I have extensive experience as a tutor, both online and in-person. I have worked with students of all ages and abilities, and am skilled at adapting my teaching style to meet the needs of each individual student. I have a strong background in a variety of subjects, including math, science, and English, and am able to break down complex concepts in a way that is easy for students to understand. In addition to my subject matter expertise, I am also a patient and supportive teacher, and am committed to helping my students succeed. Whether I am working with a struggling student who needs extra help to catch up, or an advanced student looking to get ahead, I am able to provide the guidance and support they need to reach their goals. Overall, my hands-on experience as a tutor has prepared me to be a confident and effective teacher, and I am excited to use my skills to help students succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
A mass-loaded piston/cylinder containing air is at 300 kPa, 17C with a volume of 0.25 m3, while at the stops V 1 m3. An air line, 500 kPa, 600 K, is connected by a valve that...
-
A mass-loaded piston/cylinder containing air is at 45 lbf/in 2, 60 F with a volume of 9 ft3, while at the stops V 36 ft3. An air line, 75 lbf/in 2, 1100 R, is connected by a valve....
-
A mass-loaded piston/cylinder containing air is at 45 lbf/in. 2 , 60 F with a volume of 9 ft 3 , while at the stops V = 36 ft 3 . An air line, 75 lbf./in. 2 , 1100 R is connected by a valve, as shown...
-
A trial balance was extracted from the books of V Baker, and it was found that the debit side exceeded the credit side by 40. This amount was entered in the suspense account. The following errors...
-
Describe the relationship between information security policies and an information security plan.
-
A fish rests on the bottom of a bucket of water while the bucket is being weighed on a scale. When the fish begins to swim around, does the scale reading change?
-
1.19
-
Craig Products Co. maintains the following accounts in the general ledger: Materials, Work in Process, Factory Overhead, and Accounts Payable. On June 1, the materials account had a debit balance of...
-
Sheridan Company signed a lease for an office building for a period of 13 years. Under the lease agreement, a security deposit of $10,200 is made. The deposit will be returned at the expiration of...
-
Hemming uses a perpetual inventory system. 1. Determine the costs assigned to ending inventory and to cost of goods sold using FIFO. 2. Determine the costs assigned to ending inventory and to cost of...
-
An insulated spring-loaded piston/cylinder device, shown in Fig. P4.153, is connected to an air line flowing air at 600 kPa and 700 K by a valve. Initially, the cylinder is empty and the spring force...
-
A 2-m 3 storage tank contains 95% liquid and 5% vapor by volume of liquified natural gas (LNG) at 160 K, as shown in Fig. P4.155. It may be assumed that LNG has the same properties as pure methane....
-
What is the blood vessel that transports blood to the right atrium called?
-
Based on the case, Insights Analytics: Technology for a Knowledge Management Program attached . Please explain all 8 points. Explanation of each point should be 300words . Please attach the reference...
-
When women were finally allowed to become pilots of fighter jets, engineers needed to redesign the ejection seats because they had been originally designed for men only. The ejection seats were...
-
What will be the output of the following code snippet? with open ("hello.txt", "w") as f: f.write("Hello World how are you today") with open('hello.txt', 'r') as f: data = f.readlines () for line in...
-
Assume that females have pulse rates that are normally distributed with a mean of p = 72.0 beats per minute and a standard deviation of o = 12.5 beats per minute. Complete parts (a) through (c)...
-
The proposed rates were not in the range the CEO expected given the pricing analysis. The CEO has asked the pricing actuary to verify the total projected loss cost excluding potential large storm...
-
Write the following two generic methods using bubble sort. The first method sorts the elements using the Comparable interface and the second uses the Comparator interface. public static void...
-
The diagram shows the two forces acting on a small object. Which of the following is the resultant force on the object? A. 8 N downwards B. 8 N upwards C. 2 N downwards D. 2 N upwards 3 N 5 N
-
Find the flows of exergy into and out of the feedwater heater in Problem 11.43.
-
Find the availability of the water at all the states in the steam power plant described in Problem 11.57. Assume the heat source in the boiler is at 600C and the low-temperature reservoir is at 25C....
-
How many parameters do you need to know to completely describe the Otto cycle? How about the Diesel cycle?
-
Discuss why it is important for company managers to understand and use social capital knowledge to help build social ties among their skilled knowledge workers so they can build employee loyalty...
-
Kate lives in a house close to a local university, and she traditionally has rented a garage apartment in the back of her property to students for $750 per month. Kate wants to transfer the title to...
-
Pottery Ranch Inc. has been manufacturing its own finials for its curtain rods. The company is currently operating at 100% of capacity, and variable manufacturing overhead is charged to production at...

Study smarter with the SolutionInn App


